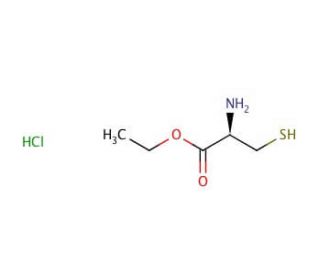
L-Cysteine ethyl ester hydrochloride (CAS 868-59-7)
ACCÈS RAPIDE AUX LIENS
Le chlorhydrate d'ester éthylique de L-cystéine est un composé qui sert de précurseur dans la synthèse des protéines et des peptides. Il joue un rôle dans la formation de liaisons disulfures au sein des protéines, contribuant ainsi à leur stabilité structurelle. Au niveau moléculaire, le chlorhydrate de L-cystéine éthyl ester participe aux réactions d'oxydoréduction, servant d'agent réducteur pour maintenir l'équilibre thiol-disulfure dans les cellules. Le chlorhydrate de L-cystéine éthyl ester agit également comme un piégeur de radicaux libres, contribuant à protéger les cellules des dommages oxydatifs. Le chlorhydrate de L-cystéine éthyl ester est impliqué dans la régulation des voies de signalisation cellulaires, influençant des processus tels que la croissance et la différenciation cellulaires. Son mécanisme d'action comprend des interactions avec des enzymes et d'autres protéines, modulant leur activité et leur fonction dans l'environnement cellulaire.
L-Cysteine ethyl ester hydrochloride (CAS 868-59-7) Références
- Structure moléculaire à l'état solide du dérivé S-nitroso du chlorhydrate d'ester éthylique de L-cystéine. | Yi, J., et al. 2005. Nitric Oxide. 12: 261-6. PMID: 15917219
- Quantification des lactones sesquiterpéniques dans les extraits de plantes Asteraceae: Évaluation de leur potentiel allergène. | Salapovic, H., et al. 2013. Sci Pharm. 81: 807-18. PMID: 24106675
- Comportement vibrationnel et structurel du chlorhydrate d'ester éthylique de (L)-cystéine à l'état solide et en solution aqueuse. | Defonsi Lestard, ME., et al. 2013. J Phys Chem A. 117: 14243-52. PMID: 24328050
- Réponses in vitro et in vivo à la bio-interaction et évaluation de la biosécurité de nouvelles nanoparticules à noyau Au-ZnTe. | Dunpall, R. and Revaprasadu, N. 2016. Toxicol Res (Camb). 5: 1078-1089. PMID: 30090413
- Identification d'une nouvelle sérine alcaline peptidase provenant de la souche FarDT du Virgibacillus natechei sp. nov. modérément halophile et son application en tant que bioadditif pour la synthèse de peptides et les formulations de détergents de lessive. | Mechri, S., et al. 2019. Biomed Res Int. 2019: 6470897. PMID: 31886235
- L'inhibition de l'activité de l'uréase par différents composés permet de mieux comprendre la modulation et l'association de l'importation de nickel bactérien et de l'uréolyse. | Svane, S., et al. 2020. Sci Rep. 10: 8503. PMID: 32444844
- Types polyvalents de systèmes d'administration de médicaments à base de polysaccharides: De la conception stratégique à la thérapie du cancer. | Sun, Y., et al. 2020. Int J Mol Sci. 21: PMID: 33271967
- Détection chiroptique de dérivés d'acides aminés par complexation hôte-invité avec le cyclo[6]aramide. | Wang, X., et al. 2021. Molecules. 26: PMID: 34279403
- Identification du L-cystéinamide comme inhibiteur puissant de la formation du dopachrome et de la synthèse de l'eumélanine médiées par la tyrosinase. | Lee, HK., et al. 2021. Antioxidants (Basel). 10: PMID: 34439449
- Systèmes d'administration de médicaments auto-émulsifiants ciblés pour restaurer la sensibilité du docétaxel dans les tumeurs résistantes. | Campani, V., et al. 2022. Pharmaceutics. 14: PMID: 35214025
- Transformation topotactique de matériaux homogènes à base d'acide phosphotungastomolybdique en catalyseur acide solide hétérogène pour la conversion des hydrates de carbone en méthylfurfural d'alkyle et en lévulinate d'alkyle. | Gupta, D., et al. 2020. RSC Adv. 10: 705-718. PMID: 35494434
- Plate-forme pour la modification orthogonale de protéines spécifiques à la N-cystéine grâce à des réactifs à base de cyclopropénone. | Istrate, A., et al. 2022. J Am Chem Soc. 144: 10396-10406. PMID: 35658467
- Nouveaux dérivés de benzothiazole en tant que ligands à cibles multiples pour le traitement de la maladie d'Alzheimer. | Hafez, DE., et al. 2023. J Enzyme Inhib Med Chem. 38: 2175821. PMID: 36789662
Informations pour la commande
| Nom du produit | Ref. Catalogue | COND. | Prix HT | QTÉ | Favoris | |
L-Cysteine ethyl ester hydrochloride, 25 g | sc-250214 | 25 g | $39.00 |
