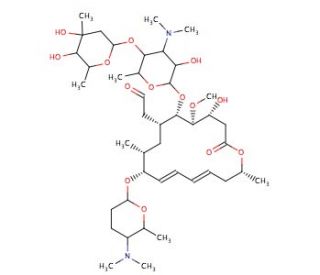
Spiramycin I (CAS 24916-50-5)
Direktverknüpfungen
Spiramycin I, ein prominentes Mitglied der Makrolid-Antibiotika, zeichnet sich durch seine große makrozyklische Lacton-Ringstruktur aus, die auf komplizierte Weise mit Zuckern verziert ist. Diese molekulare Architektur ist ausschlaggebend für seinen Wirkmechanismus, der in erster Linie auf bakterielle Ribosomen abzielt, um die Proteinsynthese zu hemmen. Durch Bindung an die 50S-Untereinheit des Ribosoms blockiert Spiramycin I wirksam den Translokationsschritt der Proteinverlängerung und stoppt so das bakterielle Wachstum. Diese Wirkungsweise ist besonders wertvoll für die Forschung, wo Spiramycin I als Werkzeug zur Untersuchung der Feinheiten der bakteriellen Proteinsynthese und der Ribosomenfunktion dient. Sein Nutzen geht über die reine antibakterielle Anwendung hinaus, da es Einblicke in die ribosomale Struktur und Funktion gewährt, die für das Verständnis der grundlegenden Prozesse des Lebens und für die Entwicklung neuer Antibiotika wichtig sind.
Spiramycin I (CAS 24916-50-5) Literaturhinweise
- Analyse von Spiramycin durch Kapillarelektrophorese. | González-Hernández, R., et al. 1999. Electrophoresis. 20: 2407-11. PMID: 10499332
- Gleichzeitige Bestimmung von Metronidazol und Spiramycin I in menschlichem Plasma, Speichel und Zahnfleischflüssigkeit mittels LC-MS/MS. | Sagan, C., et al. 2005. J Pharm Biomed Anal. 38: 298-306. PMID: 15925222
- [Deletion des Spiramycin 3-O-Acyltransferase-Gens von Streptomyces spiramyceticus F21, was zur Produktion von Spiramycin I als Hauptkomponente führt]. | Wu, LZ., et al. 2007. Sheng Wu Gong Cheng Xue Bao. 23: 612-7. PMID: 17849607
- Hydroxylierung und Hydrolyse: zwei wichtige Stoffwechselwege von Spiramycin I in der anaeroben Verdauung. | Zhu, P., et al. 2014. Bioresour Technol. 153: 95-100. PMID: 24345568
- Anaerober biologischer Abbau von Spiramycin I und Charakterisierung seiner neuen Metaboliten. | Yao, J., et al. 2017. Biosci Biotechnol Biochem. 81: 1051-1054. PMID: 28095730
- Stimulierende Effekte von Methyl-β-Cyclodextrin auf die Spiramycin-Produktion und physikalisch-chemische Charakterisierung von Nonhost@Guest-Komplexen. | Calcagnile, M., et al. 2018. ACS Omega. 3: 2470-2478. PMID: 30221219
- Struktur-Aktivitäts-Beziehungen von Spiramycinen. | Omura, S., et al. 1985. J Antimicrob Chemother. 16 Suppl A: 1-11. PMID: 3902764
- Chemische Modifikation von Spiramycinen. V. Synthese und antibakterielle Aktivität von 3'- oder 4''-de-N-Methylspiramycin I und ihrer N-substituierten Derivate. | Sano, H., et al. 1985. J Antibiot (Tokyo). 38: 186-96. PMID: 3997666
- Chemische Modifikation von Spiramycinen. VI. Synthese und antibakterielle Aktivitäten von 3,3''-Di-O-acyl-4''-O-sulfonyl und 3,3''-Di-O-acyl-4''-O-alkyl Derivaten von Spiramycin I. | Sano, M., et al. 1985. J Antibiot (Tokyo). 38: 1350-8. PMID: 4066488
- Chemische Modifikation von Spiramycinen. III. Synthese und antibakterielle Aktivitäten von 4''-Sulfonaten und 4''-Alkylethern von Spiramycin I. | Sano, H., et al. 1984. J Antibiot (Tokyo). 37: 750-9. PMID: 6469869
- Chemische Modifikation von Spiramycinen. IV. Synthese und in vitro- und in vivo-Aktivitäten von 3'',4''-Diacylaten und 3,3'',4''-Triacylaten von Spriamycin I. | Sano, H., et al. 1984. J Antibiot (Tokyo). 37: 760-72. PMID: 6469870
- Chemische Modifikation von Spiramycinen. I. Synthese der Acetalderivate von Neospiramycin I. | Sano, H., et al. 1983. J Antibiot (Tokyo). 36: 1336-44. PMID: 6643282
- Biosynthese und metabolisches Schicksal von Kohlenstoff-14-markiertem Spiramycin I. | Inoue, A., et al. 1983. J Antibiot (Tokyo). 36: 442-4. PMID: 6853373
Bestellinformation
| Produkt | Katalog # | EINHEIT | Preis | ANZAHL | Favoriten | |
Spiramycin I, 2.5 g | sc-220129 | 2.5 g | $331.00 |
