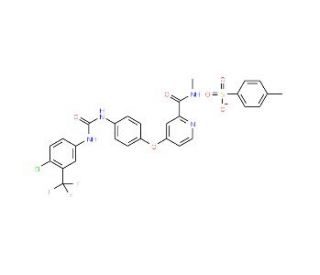
Sorafenib Tosylate (CAS 475207-59-1)
See product citations (16)
QUICK LINKS
Sorafenib Tosylate exerts broad-spectrum anticancer efficacy in vitro via targeting b-Raf, c-Raf (Raf-1), as well as several receptor tyrosine kinases involved in neovascularization and tumor progression. These tyrosibne kinases targeted by Sorafenib Tosylate include vascular endothelial growth factor receptors 2/3 (VEGFR-2/Flk-1/KDR, VEGFR-3), platelet-derived growth factor receptor-beta (PDGFR-β), Flt-3, c-KIT, FGFR-1 (Flt-2) and RET. Sorafenib Tosylate disrupts signaling pathways that are vital for tumor growth and angiogenesis, the process through which tumors develop their own blood supply. Researchers use Sorafenib Tosylate to dissect the molecular pathways that are critical for cancer cell survival and proliferation.
Sorafenib Tosylate (CAS 475207-59-1) References
- Bevacizumab, sorafenib tosylate, sunitinib and temsirolimus for renal cell carcinoma: a systematic review and economic evaluation. | Thompson Coon, J., et al. 2010. Health Technol Assess. 14: 1-184, iii-iv. PMID: 20028613
- In vitro to in vivo comparison of the substrate characteristics of sorafenib tosylate toward P-glycoprotein. | Gnoth, MJ., et al. 2010. Drug Metab Dispos. 38: 1341-6. PMID: 20413726
- A patient with advanced hepatocellular carcinoma treated with sorafenib tosylate showed massive tumor lysis with avoidance of tumor lysis syndrome. | Joshita, S., et al. 2010. Intern Med. 49: 991-4. PMID: 20519814
- Sorafenib tosylate and paclitaxel induce anti-angiogenic, anti-tumour and anti-resorptive effects in experimental breast cancer bone metastases. | Merz, M., et al. 2011. Eur J Cancer. 47: 277-86. PMID: 20863686
- Improving anti-tumor activity of sorafenib tosylate by lipid- and polymer-coated nanomatrix. | Guo, Y., et al. 2017. Drug Deliv. 24: 270-277. PMID: 28165798
- Sorafenib tosylate inhibits directly necrosome complex formation and protects in mouse models of inflammation and tissue injury. | Martens, S., et al. 2017. Cell Death Dis. 8: e2904. PMID: 28661484
- Development of a validated liquid chromatographic method for quantification of sorafenib tosylate in the presence of stress-induced degradation products and in biological matrix employing analytical quality by design approach. | Sharma, T., et al. 2018. Biomed Chromatogr. 32: e4169. PMID: 29244215
- Effect of PEGylation on assembly morphology and cellular uptake of poly ethyleneimine-cholesterol conjugates for delivery of sorafenib tosylate in hepatocellular carcinoma. | Monajati, M., et al. 2018. Bioimpacts. 8: 241-252. PMID: 30397579
- Development of Formulation Methods and Physical Characterization of Injectable Sodium Selenite Nanoparticles for the Delivery of Sorafenib tosylate. | Moni, SS., et al. 2020. Curr Pharm Biotechnol. 21: 659-666. PMID: 31886748
- Supersaturated LFCS type III self-emulsifying delivery systems of sorafenib tosylate with improved biopharmaceutical performance: QbD-enabled development and evaluation. | Sharma, T., et al. 2020. Drug Deliv Transl Res. 10: 839-861. PMID: 32415654
- Folic acid modified gold nanoparticle for targeted delivery of Sorafenib tosylate towards the treatment of diabetic retinopathy. | Dave, V., et al. 2020. Colloids Surf B Biointerfaces. 194: 111151. PMID: 32540764
- Synthesis, anticancer activity, and β-lactoglobulin binding interactions of multitargeted kinase inhibitor sorafenib tosylate (SORt) using spectroscopic and molecular modelling approaches. | Tanzadehpanah, H., et al. 2021. Luminescence. 36: 117-128. PMID: 32725773
- Self Nanoemulsifying Drug Delivery System of Sorafenib Tosylate: Development and In Vivo Studies. | Sandhya, P., et al. 2020. Pharm Nanotechnol. 8: 471-484. PMID: 33069205
- Polymeric Nanocarriers for Effective Synergistic Action of Sorafenib Tosylate and Gold-sensitized Gamma Radiation Against HepG2 Cells. | Sukkar, F., et al. 2021. Int J Nanomedicine. 16: 8309-8321. PMID: 34992367
- Formulating Ternary Inclusion Complex of Sorafenib Tosylate Using β-Cyclodextrin and Hydrophilic Polymers: Physicochemical Characterization and In Vitro Assessment. | Donthi, MR., et al. 2022. AAPS PharmSciTech. 23: 254. PMID: 36109473
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sorafenib Tosylate, 100 mg | sc-357801 | 100 mg | $104.00 | |||
Sorafenib Tosylate, 1 g | sc-357801A | 1 g | $312.00 |
