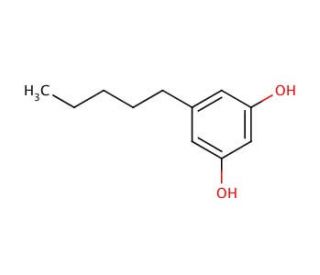
Olivetol (CAS 500-66-3)
Produktreferenzen ansehen (2)
Direktverknüpfungen
Olivetol, eine organische Verbindung, wird häufig in der synthetischen Chemie sowie in der biochemischen Forschung eingesetzt. Seine strukturelle Ähnlichkeit zu bestimmten natürlichen Cannabinoiden macht es zu einem wertvollen Werkzeug für die Untersuchung des Endocannabinoid-Systems. Biochemiker erforschen seine Rolle in biosynthetischen Pfaden, insbesondere solchen, die zur Produktion von Polyketiden führen, einer Klasse sekundärer Metaboliten mit vielfältigen biologischen Aktivitäten. Darüber hinaus umfasst die Forschung zu Olivetol auch die Untersuchung seines Potenzials als Ligand für Cannabinoid-Rezeptoren, was dazu beiträgt, die Rezeptor-Liganden-Interaktionen und Signalwege weiter zu verstehen.
Olivetol (CAS 500-66-3) Literaturhinweise
- Herstellung und Bewertung eines molekular geprägten Polymers von Olivetol für die Festphasenextraktion. | Jin, Y., et al. 2013. Se Pu. 31: 587-95. PMID: 24063201
- Charakterisierung der strukturellen Determinanten, die für eine starke, auf einem Mechanismus basierende Hemmung des menschlichen Cytochroms P450 1A1 durch Cannabidiol erforderlich sind. | Yamaori, S., et al. 2014. Chem Biol Interact. 215: 62-8. PMID: 24667653
- Die hemmende Wirkung von Cannabinoiden, den aktiven Bestandteilen von Cannabis sativa L., auf die Thrombozytenaggregation bei Menschen und Kaninchen. | Formukong, EA., et al. 1989. J Pharm Pharmacol. 41: 705-9. PMID: 2575149
- Neuartige Δ8-Tetrahydrocannabinol-Verdampfer enthalten nicht gekennzeichnete Verunreinigungen, unbeabsichtigte Nebenprodukte der chemischen Synthese und Schwermetalle. | Meehan-Atrash, J. and Rahman, I. 2022. Chem Res Toxicol. 35: 73-76. PMID: 34889611
- Stereoselektive Synthese von nicht-psychotischem natürlichem Cannabidiol und seinen unnatürlichen/Terpenyl/Schwanz-modifizierten Analoga. | Anand, R., et al. 2022. J Org Chem. 87: 4489-4498. PMID: 35289168
- Synthetische Strategien für seltene Cannabinoide, die aus Cannabis sativa gewonnen werden. | Nguyen, GN., et al. 2022. J Nat Prod. 85: 1555-1568. PMID: 35648593
- Carbonylfalleigenschaften von 5-Alkylresorcinolen. | Zamora, R. and Hidalgo, FJ. 2022. Food Chem. 393: 133372. PMID: 35661596
- Simulierte Mikrogravitation trug zur Veränderung der Kallogeneseleistung und der Produktion von Sekundärmetaboliten bei CannabisIndica bei. | Darigh, F., et al. 2022. Plant Physiol Biochem. 186: 157-168. PMID: 35849945
- Elektrochemischer Nachweis von Olivetol auf der Grundlage einer mit einem Poly(L-Serin)-Film beschichteten Kupferoxid-modifizierten Kohlenstoffpastelektrode (p-L-Serin/CuO/CPE). | You, Z., et al. 2022. Nanomaterials (Basel). 13: PMID: 36615980
- Abfangen von Malondialdehyd durch Lebensmittelphenole. | Zamora, R., et al. 2023. Food Chem. 417: 135915. PMID: 36933433
- Analyse von Olivetol in Kaninchenserum durch Hochleistungsflüssigkeitschromatographie. | Rauls, D. and Penney, LL. 1982. J Chromatogr. 234: 500-2. PMID: 7056834
- Auswirkungen von Cannabinoiden auf die makromolekulare Synthese und Replikation von kultivierten Lymphozyten. | Nahas, GG., et al. 1977. Fed Proc. 36: 1748-52. PMID: 844617
- Quantenmechanische und experimentelle Oxidationsstudien von Pentadecylresorcin, Olivetol, Orcinol und Resorcinol. | Hładyszowski, J., et al. 1998. Free Radic Res. 28: 359-68. PMID: 9684980
Bestellinformation
| Produkt | Katalog # | EINHEIT | Preis | ANZAHL | Favoriten | |
Olivetol, 1 g | sc-236251 | 1 g | $58.00 | |||
Olivetol, 10 g | sc-236251A | 10 g | $214.00 | |||
Olivetol, 100 g | sc-236251B | 100 g | $1047.00 | |||
Olivetol, 1 kg | sc-236251C | 1 kg | $3648.00 | |||
Olivetol, 5 kg | sc-236251D | 5 kg | $14052.00 |
