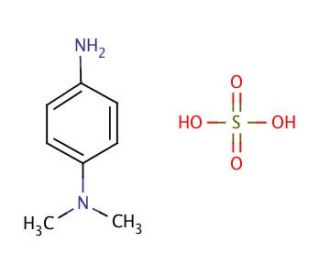
N,N-Dimethyl-p-phenylenediamine sulfate salt (CAS 536-47-0)
ACCÈS RAPIDE AUX LIENS
Le sel de sulfate de N,N-diméthyl-p-phénylènediamine (DMPD) occupe une place importante en tant que réactif largement utilisé dans le domaine de la recherche scientifique. Cette poudre cristalline blanche sert d'agent sulfonant et trouve de nombreuses applications dans diverses expériences de laboratoire. La polyvalence du sel de sulfate de N,N-diméthyl-p-phénylènediamine lui permet de remplir un large éventail de rôles, notamment en matière de synthèse, d'analyse et d'expérimentation biologique. Dans le domaine de la recherche scientifique, le sel de sulfate de N,N-Diméthyl-p-phénylènediamine joue un rôle essentiel. Son application en tant qu'agent sulfonant permet la synthèse de divers composés, tels que les colorants et les polymères. En outre, il trouve son utilité dans l'analyse des composés, servant de détecteur fiable des groupes d'acides sulfoniques. Dans le cadre d'expériences biologiques, le sel de sulfate de N,N-Diméthyl-p-phénylènediamine s'avère inestimable pour identifier des enzymes et des protéines spécifiques. La fonction clé du sel de sulfate de N,N-Diméthyl-p-phénylènediamine réside dans ses propriétés sulfonantes, qui lui permettent de réagir avec d'autres composés et de générer des groupes d'acide sulfonique. Cette réaction est extrêmement importante pour la synthèse de divers composés, car le groupe acide sulfonique peut servir de catalyseur pour des réactions ultérieures. De plus, le sel de sulfate de N,N-Diméthyl-p-phénylènediamine permet de détecter les groupes d'acide sulfonique dans d'autres composés, ce qui élargit encore ses capacités d'analyse.
N,N-Dimethyl-p-phenylenediamine sulfate salt (CAS 536-47-0) Références
- Contributions eucaryotes et procaryotes à la synthèse du sulfure d'hydrogène colique. | Flannigan, KL., et al. 2011. Am J Physiol Gastrointest Liver Physiol. 301: G188-93. PMID: 21474649
- Sulfure d'hydrogène et résolution de l'inflammation aiguë: Une étude comparative utilisant une nouvelle sonde fluorescente. | Dufton, N., et al. 2012. Sci Rep. 2: 499. PMID: 22787557
- Effets bénéfiques de la diminution de la production de sulfure d'hydrogène ou de monoxyde de carbone sur l'hypertension et les lésions rénales induites par le retrait du NO. | Wesseling, S., et al. 2015. Br J Pharmacol. 172: 1607-19. PMID: 24597655
- Production géochimique d'espèces réactives de l'oxygène à partir de Fe biogéochimiquement réduit. | Murphy, SA., et al. 2014. Environ Sci Technol. 48: 3815-21. PMID: 24597860
- La fermentation associe des bactéries chloroflexi et sulfato-réductrices à des cyanobactéries dans des tapis microbiens hypersalins. | Lee, JZ., et al. 2014. Front Microbiol. 5: 61. PMID: 24616716
- Estimation de la production de sulfure d'hydrogène bactérien in vitro. | Basic, A., et al. 2015. J Oral Microbiol. 7: 28166. PMID: 26130377
- Le glutathion réduit est un médiateur de la résistance à la toxicité du H2S chez les streptocoques oraux. | Ooi, XJ. and Tan, KS. 2016. Appl Environ Microbiol. 82: 2078-2085. PMID: 26801579
- Nouvelles approches pour atteindre un niveau élevé de production d'enzymes chez Streptomyces lividans. | Sevillano, L., et al. 2016. Microb Cell Fact. 15: 28. PMID: 26846788
- Effets du sélénium alimentaire sur l'inflammation et le sulfure d'hydrogène dans le tractus gastro-intestinal des poulets. | Wu, C., et al. 2016. Biol Trace Elem Res. 174: 428-435. PMID: 27178167
- Expression et activité des enzymes génératrices de sulfure d'hydrogène dans les macrophages murins stimulés par le lipopolysaccharide et l'interféron-γ. | Bronowicka-Adamska, P., et al. 2019. Mol Biol Rep. 46: 2791-2798. PMID: 30859447
- Nouvelles applications de l'acétate de plomb et des méthodes de cytométrie en flux pour la détection des molécules contenant du soufre. | Anishchenko, E., et al. 2019. Methods Protoc. 2: PMID: 31164595
- Formation de sulfure d'hydrogène dans un modèle expérimental de pancréatite aiguë. | Bronowicka-Adamska, P., et al. 2019. Acta Biochim Pol. 66: 611-618. PMID: 31893496
- La thérapie combinée à base de nanoparticules a montré une activité supérieure contre les pathogènes bactériens multirésistants par rapport aux médicaments libres. | Brar, A., et al. 2022. Nanomaterials (Basel). 12: PMID: 35808015
Informations pour la commande
| Nom du produit | Ref. Catalogue | COND. | Prix HT | QTÉ | Favoris | |
N,N-Dimethyl-p-phenylenediamine sulfate salt, 25 g (Out of Stock: Availability 1/20/26) | sc-250513 | 25 g | $54.00 | |||
| US: (En rupture de stock: Disponibilité Janvier 20, 2026) | ||||||
