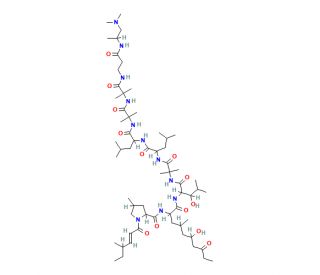
Molecular structure of Leucinostatin A, CAS Number: 76600-38-9
Leucinostatin A (CAS 76600-38-9)
See product citations (1)
Alternate Names:
Tryanocidin; Paecilotoxin A
Application:
Leucinostatin A is a broadly bioactive component produced by Paecilomyces lilacinus.
CAS Number:
76600-38-9
Purity:
≥95%
Molecular Weight:
1218.61
Molecular Formula:
C62H111N11O13
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Leucinostatin A is the major component of an atypical nonapeptide complex produced by Paecilomyces lilacinus, first reported in 1973. Leucinostatins display broad bioactivity against Gram positive bacteria, fungi, plants and tumour cell lines. Leucinostatin A inhibits respiration by uncoupling oxidative phosphorylation and is potentiated by inhibitors such as venturicidin and oligomycin. More recently, interest in leucinostatin has focused on understanding its activity as an insulin-like growth factor I regulator, an ionophore, inhibitor of cell surface expression of viral glycoproteins and its anti- trypanosomal activity.
Leucinostatin A (CAS 76600-38-9) References
- Microbiological and densitometric TLC analyses for peptides in liposomes. | Ricci, M., et al. 2001. J Pharm Biomed Anal. 25: 903-12. PMID: 11377073
- Leucinostatin-A loaded nanospheres: characterization and in vivo toxicity and efficacy evaluation. | Ricci, M., et al. 2004. Int J Pharm. 275: 61-72. PMID: 15081138
- Leucinostatin A inhibits prostate cancer growth through reduction of insulin-like growth factor-I expression in prostate stromal cells. | Kawada, M., et al. 2010. Int J Cancer. 126: 810-8. PMID: 19795463
- The crystal and molecular structure of the alpha-helical nonapeptide antibiotic leucinostatin A. | Cerrini, S., et al. 1989. Biopolymers. 28: 409-20. PMID: 2720117
- Catalytic Asymmetric Total Synthesis and Stereochemical Revision of Leucinostatin A: A Modulator of Tumor-Stroma Interaction. | Abe, H., et al. 2017. Chemistry. 23: 11792-11796. PMID: 28703358
- Chemical Studies on Bioactive Natural Products Directed toward Development of Novel Antiinfective and Anticancer Medicines. | Watanabe, T. 2019. Chem Pharm Bull (Tokyo). 67: 620-631. PMID: 31257316
- Inhibition of mitochondria ATP synthase suppresses prostate cancer growth through reduced insulin-like growth factor-1 secretion by prostate stromal cells. | Ohishi, T., et al. 2020. Int J Cancer. 146: 3474-3484. PMID: 32144767
- Catalytic Asymmetric Total Synthesis of Leucinostatin A. | Watanabe, T., et al. 2021. Chem Rec. 21: 175-187. PMID: 33107684
- Antiprotozoal Structure-Activity Relationships of Synthetic Leucinostatin Derivatives and Elucidation of their Mode of Action. | Brand, M., et al. 2021. Angew Chem Int Ed Engl. 60: 15613-15621. PMID: 33730410
- Structure activity studies on chemically modified homologues of the antibiotic phytotoxic leucinostatin A. | Vertuani, G., et al. 1995. J Antibiot (Tokyo). 48: 254-60. PMID: 7730161
- The nonapeptide leucinostatin A acts as a weak ionophore and as an immunosuppressant on T lymphocytes. | Csermely, P., et al. 1994. Biochim Biophys Acta. 1221: 125-32. PMID: 8148389
- Novel blockade of cell surface expression of virus glycoproteins by leucinostatin A. | Muroi, M., et al. 1996. J Antibiot (Tokyo). 49: 1119-26. PMID: 8982341
- Glucosylation of the peptide leucinostatin A, produced by an endophytic fungus of European yew, may protect the host from leucinostatin toxicity. | Strobel, GA. and Hess, WM. 1997. Chem Biol. 4: 529-36. PMID: 9263641
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Leucinostatin A, 0.5 mg | sc-391623 | 0.5 mg | $208.00 |
