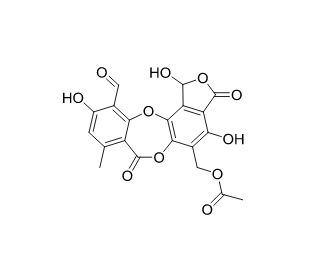
Galbinic acid (CAS 56691-88-4)
ENLACES RÁPIDOS
El ácido galbínico, un derivado flavonoide, es un compuesto natural que se encuentra en varias especies vegetales, en particular las pertenecientes a la familia Fabaceae. Se caracteriza por un marco estructural distinto que incluye un grupo hidroxilo fenólico, lo que contribuye significativamente a sus propiedades químicas y a su relevancia biológica en entornos de investigación no médicos. Como producto químico de investigación, el ácido galbínico se ha utilizado ampliamente en estudios centrados en la biología y la ecología de las plantas, especialmente para comprender los mecanismos de defensa de las plantas frente a factores de estrés ambiental. Su mecanismo de acción implica principalmente actividades antioxidantes, en las que elimina los radicales libres y protege los tejidos vegetales del daño oxidativo causado por la radiación UV y los ataques patógenos. Esta propiedad antioxidante hace del ácido galbínico una valiosa herramienta en los estudios fisiológicos de las plantas, ya que ayuda a explicar cómo éstas hacen frente al estrés oxidativo y se adaptan a su entorno. Además, la función del ácido galbínico en las vías de señalización que regulan el crecimiento y las respuestas de defensa de las plantas ha sido objeto de investigación botánica, lo que permite comprender mejor las complejas interacciones dentro de las células vegetales y entre las plantas y su entorno. Estos estudios contribuyen a una comprensión más amplia de la biología vegetal y ayudan a desarrollar estrategias para la mejora de los cultivos y las prácticas agrícolas sostenibles.
Galbinic acid (CAS 56691-88-4) Referencias
- Una nueva depsidona y actividades antibacterianas de compuestos de Usnea undulata Stirton. | Sultana, N. and Afolayan, AJ. 2011. J Asian Nat Prod Res. 13: 1158-64. PMID: 22115039
- Estudio taxonómico del género myelochroa en Corea del Sur. | Jayalal, U., et al. 2012. Mycobiology. 40: 217-24. PMID: 23323045
- Nuevos conocimientos sobre el agregado Usnea cornuta (Parmeliaceae, lichenized Ascomycota): El análisis molecular revela una elevada diversidad genética correlacionada con la química. | Gerlach, ADCL., et al. 2019. Mol Phylogenet Evol. 131: 125-137. PMID: 30385309
- Actividad Antimicrobiana del Ácido Divaricatico Aislado del Liquen Evernia mesomorpha contra el Staphylococcus aureus Resistente a la Meticilina. | Oh, JM., et al. 2018. Molecules. 23: PMID: 30477128
- Los extractos de Flavoparmelia sp. inhiben la diferenciación de los osteoclastos mediada por el ligando activador del receptor del factor nuclear-κB. | Kim, KJ., et al. 2019. J Bone Metab. 26: 113-121. PMID: 31223608
- Depsidonas y Difeniléteres Derivados del Ácido Salazínico con Actividad Inhibidora de la α-Glucosidasa del Liquen Parmotrema dilatatum. | Devi, AP., et al. 2020. Planta Med. 86: 1216-1224. PMID: 32819010
- Cribado de metabolitos secundarios criptogámicos como inhibidores putativos de la proteasa principal del SARS-CoV-2 y del dominio de unión ribosomal de la glicoproteína espiga mediante enfoques de acoplamiento molecular y dinámica molecular. | Prateeksha, G., et al. 2021. J Mol Struct. 1240: 130506. PMID: 33967344
- Una aplicación sostenible para la extracción de metabolitos liquénicos de Usnea cornuta: metabolómica no dirigida y actividad antioxidante. | Castañeta, G., et al. 2023. Nat Prod Res. 37: 2076-2082. PMID: 36008873
- Estudio comparativo de las actividades antimicrobianas y los perfiles metabólicos de cinco especies de Usnea de Filipinas. | Dela Cruz, TEE., et al. 2023. J Fungi (Basel). 9: PMID: 37998922
- Estructura del ácido galbínico. Una depsidona del liquen Usnea undulata. | Elix, John A and Udomsri Engkaninan. 1975. Australian journal of chemistry. 28.8: 1793-1797.
Información sobre pedidos
| Nombre del producto | Número de catálogo | UNIDAD | Precio | CANTIDAD | Favoritos | |
Galbinic acid, 1 mg (Out of Stock: Availability 6/8/26) | sc-506579 | 1 mg | $163.00 | |||
| EE.UU: (Agotado: Disponibilidad Junio 8, 2026) | ||||||
