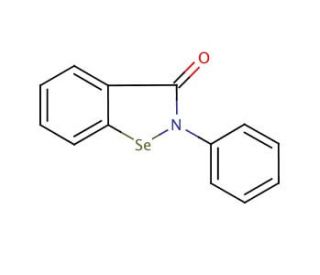
Structure moléculaire de Ebselen, Numéro CAS: 60940-34-3
Ebselen (CAS 60940-34-3)
Voir les citations produits (5)
Noms alternatifs:
2-Phenyl-1,2-benzisoselenazol-3(2H)-one; Harmokisane
Application(s):
Ebselen est un modificateur de cystéine qui inhibe la 5-LO, la 15-LO, la Cox, la NADPH et la NADPH oxydase.
Numéro CAS:
60940-34-3
Pureté:
≥98%
Masse Moléculaire:
274.18
Formule Moléculaire:
C13H9NOSe
Information supplémentaire:
Il s'agit d'une marchandise dangereuse pour le transport qui peut faire l'objet de frais d'expédition supplémentaires.
Pour la Recherche Uniquement. Non conforme pour le Diagnostic ou pour une Utilisation Thérapeutique.
* Consulter le Certificat d'Analyses pour les données spécifiques à un lot (incluant la teneur en eau).
ACCÈS RAPIDE AUX LIENS
Informations pour la commande
Références bibliographiques
Description
Information Technique
Données de Sécurité
SDS & Certificat d’Analyses
L'ebselen est un composé organosélénié qui présente des propriétés anti-inflammatoires et antioxydantes. L'ebselen modifie les résidus de cystéine et inhibe les enzymes 5-LO (5-lipoxygénase) et 15-LO en l'absence de glutathion. L'ebselen inhibe également la protéine kinase C, les cyclooxygénases Cox-1 et Cox-2, la NOS (oxyde nitrique synthase), la GST (glutathion S-transférase), l'IDO (indoleamine-pyrrole 2,3-dioxygénase), la NADPH (nicotinamide adénine dinucléotide phosphate oxydase) et la NADPH oxydase. L'oxydation des lipoprotéines de faible densité par les radicaux libres est supprimée par l'action antioxydante de l'Ebselen.
Ebselen (CAS 60940-34-3) Références
- Ebselen: un catalyseur dépendant de la thiorédoxine réductase pour la réduction de la quinone alpha-tocophérol. | Fang, J., et al. 2005. Toxicol Appl Pharmacol. 207: 103-9. PMID: 15979675
- Effet inhibiteur de l'ebselen sur l'activité de l'acétylcholinestérase cérébrale in vitro: cinétique et réversibilité de l'inhibition. | Martini, F., et al. 2015. Curr Pharm Des. 21: 920-4. PMID: 25312723
- Réutilisation de l'Auranofin, de l'Ebselen et du PX-12 en tant qu'agents antimicrobiens ciblant le système de la thiorédoxine. | May, HC., et al. 2018. Front Microbiol. 9: 336. PMID: 29556223
- La petite molécule ebselen, réactive à la cystéine, facilite la maturation efficace de la SOD1. | Capper, MJ., et al. 2018. Nat Commun. 9: 1693. PMID: 29703933
- Ciblage de la transpeptidase LdtMt2 de Mycobacterium tuberculosis par des inhibiteurs réagissant à la cystéine, dont l'ebselen. | de Munnik, M., et al. 2019. Chem Commun (Camb). 55: 10214-10217. PMID: 31380528
- L'ebselen atténue l'ototoxicité induite par la tobramycine chez la souris. | Gu, R., et al. 2021. J Cyst Fibros. 20: 271-277. PMID: 32147183
- L'ebselen améliore les lésions d'ischémie-reperfusion rénale en renforçant l'autophagie chez les rats. | Wu, Y., et al. 2022. Mol Cell Biochem. 477: 1873-1885. PMID: 35338455
- Activité antibactérienne de l'Ebselen. | Maślanka, M. and Mucha, A. 2023. Int J Mol Sci. 24: PMID: 36675123
- Actions moléculaires de l'ebselen, un antioxydant anti-inflammatoire. | Schewe, T. 1995. Gen Pharmacol. 26: 1153-69. PMID: 7590103
- Ebselen: un imitateur de la glutathion peroxydase. | Sies, H. 1994. Methods Enzymol. 234: 476-82. PMID: 7808321
- Effets de l'ebselen et du probucol sur les modifications oxydatives des lipides et des protéines des lipoprotéines de faible densité induites par les radicaux libres. | Noguchi, N., et al. 1994. Biochim Biophys Acta. 1213: 176-82. PMID: 8025128
- Forte inhibition des lipoxygénases de mammifères par le composé séléno-organique anti-inflammatoire ebselen en l'absence de glutathion. | Schewe, C., et al. 1994. Biochem Pharmacol. 48: 65-74. PMID: 8043032
- Interaction de l'ebselen avec la glutathion S-transférase et la papaïne in vitro. | Nikawa, T., et al. 1994. Biochem Pharmacol. 47: 1007-12. PMID: 8147899
- Ebselen, un composé séléno-organique imitant la glutathion peroxydase. | Sies, H. 1993. Free Radic Biol Med. 14: 313-23. PMID: 8458589
- L'ebselen comme imitateur de la glutathion peroxydase et comme piégeur de peroxynitrite. | Sies, H. and Masumoto, H. 1997. Adv Pharmacol. 38: 229-46. PMID: 8895811
Inhibiteur de:
α-defensin 2, α-defensin 6, β-Arrestin-2, β-defensin 130, β-defensin 131, 12-LO, 15-LO, 2810408P10Rik, 5-LO, 9130014G24Rik, A630033E08Rik, AATM, AKR1B10, AKR1C21, ALOXE3, apoL7b, AsnRS, ASTE1, AU017455, AZ2, CABC1, CCL (C-C motif chemokine ligand), ChM-I, CHMP7, CNOX, Cox, Cox-2, Cyp4f14, DECR1, DPP3, EG665927, FAM105B, GGCT, gp91-phox, gp91phox, GPx-1, GPx-2, Grx2, GSH-2, GST, GSTA2, GSTK1, GSTP1, Hep A, Iba1, IDO, IMP-3, IMPA1, IMPA3, KRTAP12-2, LCE1E, LCE2B, Lipid A, Ly6G6c, LYZL2, MCP-1, Mcsp, MOX-1, MsrA, MsrB3, MTERFD3, NAIP2, NIBP, NOS, NOXA1, NOXO1, NOXRED1, NSP3, Nucleoredoxin, OASL2, p22-phox, p40-phox, p47-phox, Pex7p, PHYHD1, PKC, Prdx6-rs2, PRG-3, PRX II, PRX1, PRX2, PSTK, Rad27, RADIL, RDH11, Ros, SBP-2, SDR39U1, SELB, selenocysteine lyase, Selenoprotein I, Selenoprotein K, Selenoprotein M marker, Selenoprotein S, Selenoprotein V, Selenoprotein W, SEP15, SEPHS1, SEPHS2, Ser/Thr Protein Kinase, SMNDC1, SPLUNC2, Streptolysin, Svs7, TATDN2, Tau, TMC8, TRPC4AP, Trx-2, TrxR1, TrxR2, TXNDC10, TXNDC13, TXNDC14, TXNL6, UGRP2, ZIP3, ZNF191, ZNF202, ZNF256, ZNF407, ZNF552, ZNF555, ZNF682, ZNF701, et ZNF708.Activateur de:
9430025M13Rik, AKR1B7, GGCT, GPx-4, GPx-6, GPx-8, KSP37, Miner2, Mox1, PDIR, SELENBP2, selenocysteine lyase, Selenoprotein M, Selenoprotein N, Selenoprotein O, Selenoprotein R, Sptrx-1, Trx, et TrxR3.Informations pour la commande
| Nom du produit | Ref. Catalogue | COND. | Prix HT | QTÉ | Favoris | |
Ebselen, 1 mg | sc-200740B | 1 mg | $33.00 | |||
Ebselen, 25 mg | sc-200740 | 25 mg | $136.00 | |||
Ebselen, 100 mg | sc-200740A | 100 mg | $458.00 |
