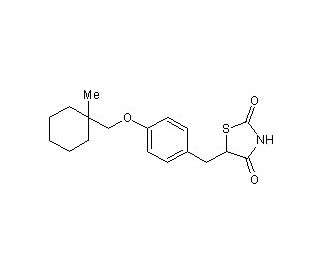
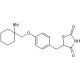
Ciglitazone (CAS 74772-77-3)
See product citations (10)
QUICK LINKS
Ciglitazone is primarily utilized in research studies focused on the mechanisms of insulin sensitization and glucose uptake regulation. In the laboratory, ciglitazone has been employed to investigate its action on peroxisome proliferator-activated receptor gamma (PPARγ), which plays a significant role in adipocyte differentiation and lipid metabolism. Researchers have explored how ciglitazone influences gene expression related to glucose and lipid metabolism, providing insights into the potential pathways through which insulin sensitivity is enhanced. Moreover, studies involving ciglitazone have contributed to understanding the metabolic effects of PPARγ agonists in various cell types, including muscle and liver cells. This research has implications for understanding metabolic diseases at a molecular level, helping to elucidate how changes in gene expression and enzyme activity can affect overall metabolic homeostasis.
Ciglitazone (CAS 74772-77-3) References
- Thiazolidinediones--the new insulin enhancers. | Jha, RJ. 1999. Clin Exp Hypertens. 21: 157-66. PMID: 10052651
- Peroxisome proliferator-activated receptor gamma ligands are potent inhibitors of angiogenesis in vitro and in vivo. | Xin, X., et al. 1999. J Biol Chem. 274: 9116-21. PMID: 10085162
- The peroxisome proliferator-activated receptor gamma (PPARgamma) ligands 15-deoxy-Delta12,14-prostaglandin J2 and ciglitazone induce human B lymphocyte and B cell lymphoma apoptosis by PPARgamma-independent mechanisms. | Ray, DM., et al. 2006. J Immunol. 177: 5068-76. PMID: 17015690
- 15-deoxy-Delta12,14-prostaglandin J2 (15d-PGJ2) and ciglitazone modulate Staphylococcus aureus-dependent astrocyte activation primarily through a PPAR-gamma-independent pathway. | Phulwani, NK., et al. 2006. J Neurochem. 99: 1389-1402. PMID: 17074064
- Inhibitory effect of meloxicam, a selective cyclooxygenase-2 inhibitor, and ciglitazone, a peroxisome proliferator-activated receptor gamma ligand, on the growth of human ovarian cancers. | Xin, B., et al. 2007. Cancer. 110: 791-800. PMID: 17582802
- Diverse cardioprotective signaling mechanisms of peroxisome proliferator-activated receptor-gamma ligands, 15-deoxy-Delta12,14-prostaglandin J2 and ciglitazone, in reperfusion injury: role of nuclear factor-kappaB, heat shock factor 1, and Akt. | Zingarelli, B., et al. 2007. Shock. 28: 554-63. PMID: 17589386
- Pro-MMP-2 activation by the PPARgamma agonist, ciglitazone, induces cell invasion through the generation of ROS and the activation of ERK. | Kim, KH., et al. 2007. FEBS Lett. 581: 3303-10. PMID: 17597617
- Rosiglitazone stimulates adipogenesis and decreases osteoblastogenesis in human mesenchymal stem cells. | Benvenuti, S., et al. 2007. J Endocrinol Invest. 30: RC26-30. PMID: 17993761
- Substituted dihydrobenzopyran and dihydrobenzofuran thiazolidine-2,4-diones as hypoglycemic agents. | Clark, DA., et al. 1991. J Med Chem. 34: 319-25. PMID: 1992133
- A protective role of ciglitazone in ox-LDL-induced rat microvascular endothelial cells via modulating PPARγ-dependent AMPK/eNOS pathway. | Xu, L., et al. 2015. J Cell Mol Med. 19: 92-102. PMID: 25388834
- Ciglitazone-a human PPARγ agonist-disrupts dorsoventral patterning in zebrafish. | Cheng, V., et al. 2019. PeerJ. 7: e8054. PMID: 31741801
- Ciglitazone, a new hypoglycemic agent. I. Studies in ob/ob and db/db mice, diabetic Chinese hamsters, and normal and streptozotocin-diabetic rats. | Chang, AY., et al. 1983. Diabetes. 32: 830-8. PMID: 6354790
- Studies on antidiabetic agents. II. Synthesis of 5-[4-(1-methylcyclohexylmethoxy)-benzyl]thiazolidine-2,4-dione (ADD-3878) and its derivatives. | Sohda, T., et al. 1982. Chem Pharm Bull (Tokyo). 30: 3580-600. PMID: 7160012
- [[omega-(Heterocyclylamino)alkoxy]benzyl]-2,4-thiazolidinediones as potent antihyperglycemic agents. | Cantello, BC., et al. 1994. J Med Chem. 37: 3977-85. PMID: 7966158
- The structure-activity relationship between peroxisome proliferator-activated receptor gamma agonism and the antihyperglycemic activity of thiazolidinediones. | Willson, TM., et al. 1996. J Med Chem. 39: 665-8. PMID: 8576907
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ciglitazone, 5 mg | sc-200902 | 5 mg | $104.00 | |||
Ciglitazone, 25 mg | sc-200902A | 25 mg | $428.00 |
