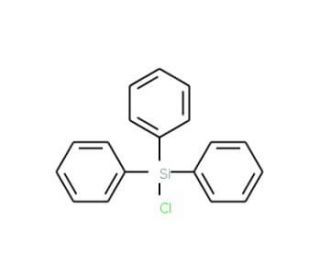
Chlorotriphenylsilane (CAS 76-86-8)
QUICK LINKS
Chlorotriphenylsilane, often utilized in the realm of synthetic organic chemistry, plays a pivotal role as a reagent in the introduction of phenyl groups into various molecular frameworks. Its mechanism of action is rooted in its ability to undergo nucleophilic substitution reactions, where the chloro group is replaced by nucleophiles, thus facilitating the formation of diverse organosilicon compounds. This capacity makes it an invaluable tool in the synthesis of complex organic molecules, enabling researchers to tailor the physicochemical properties of these molecules for specific applications. Additionally, chlorotriphenylsilane serves as a stepping stone in the creation of cross-coupling reagents and silicon-based protecting groups, further underscoring its significance in the advancement of organic synthesis methodologies. Its role extends beyond mere functionalization, as it aids in the strategic manipulation of molecular architecture, thereby contributing to the exploration and development of novel compounds with potential utility in various research domains.
Chlorotriphenylsilane (CAS 76-86-8) References
- Synthesis and structure of K+ [iPrN=C=P]-, a 1-aza-3lambda3-phospha-3-allenide. | Becker, G., et al. 2005. Inorg Chem. 44: 3080-6. PMID: 15847412
- Observation of internal electron transfer in bulky allyl ytterbium complexes with substituted terpyridine ligands. | White, RE., et al. 2006. Inorg Chem. 45: 7004-9. PMID: 16903760
- Bromo-triphenyl-silane. | Steinert, H., et al. 2008. Acta Crystallogr Sect E Struct Rep Online. 64: o880. PMID: 21202364
- New host material for high-performance blue phosphorescent organic electroluminescent devices. | Bin, JK., et al. 2012. Adv Mater. 24: 2911-5. PMID: 22549891
- σ-Bond electron delocalization of branched oligogermanes and germanium containing oligosilanes. | Hlina, J., et al. 2014. Inorganica Chim Acta. 422: 120-133. PMID: 25431502
- Unintentional PCB in chlorophenylsilanes as a source of contamination in environmental samples. | Anezaki, K. and Nakano, T. 2015. J Hazard Mater. 287: 111-7. PMID: 25634145
- Transition-Metal-Free Cross-Coupling of Benzothiophenes and Styrenes in a Stereoselective Synthesis of Substituted (E,Z)-1,3-Dienes. | Šiaučiulis, M., et al. 2019. Angew Chem Int Ed Engl. 58: 8779-8783. PMID: 30964596
- Persistent Digermenes with Acyl and α-Chlorosilyl Functionalities. | Klemmer, L., et al. 2019. Chemistry. 25: 12187-12195. PMID: 31306508
- Extending the vibrational lifetime of azides with heavy atoms. | Chalyavi, F., et al. 2020. Phys Chem Chem Phys. 22: 18007-18013. PMID: 32749405
- Conducting Silicone-Based Polymers and Their Application. | Sołoducho, J., et al. 2021. Molecules. 26: PMID: 33916125
- Oxidation of 1-O-(alk-1-enyl)-2,3-di-O-acylglycerols: models for plasmalogen oxidation. | Foglia, TA., et al. 1988. Lipids. 23: 430-4. PMID: 3412122
- Cyano(triphenylsilyl)phosphanide as a Building Block for P,C,N Conjugated Molecules. | Le Corre, G., et al. 2021. Angew Chem Int Ed Engl. 60: 24817-24822. PMID: 34463413
- Calix[6]arene-Based [3]Rotaxanes as Prototypes for the Template Synthesis of Molecular Capsules. | Cester Bonati, F., et al. 2023. Molecules. 28: PMID: 36677647
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Chlorotriphenylsilane, 5 g | sc-239538 | 5 g | $21.00 |
