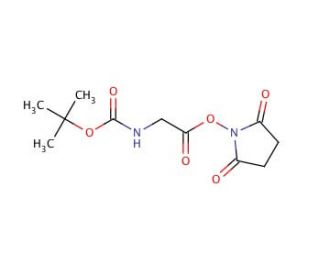
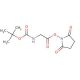
Molecular structure of Boc-Gly-OSu, CAS Number: 3392-07-2
Boc-Gly-OSu (CAS 3392-07-2)
Alternate Names:
Boc-glycine N-hydroxysuccinimide ester
CAS Number:
3392-07-2
Purity:
99%
Molecular Weight:
272.25
Molecular Formula:
C11H16N2O6
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Boc-Gly-OSu is a synthetic compound consisting of three components: Boc (tert-butyloxycarbonyl), Gly (glycine), and OSu (sulfamoylbenzamide). It serves as a valuable reagent in organic synthesis, enabling the production of a diverse range of compounds. The mechanism of action of Boc-Gly-OSu relies on its capacity to form a stable amide bond with sulfamoylbenzamide. This amide bond arises from the interaction between the carboxyl group of Boc and the amino group of OSu. The remarkable stability of this amide bond makes Boc-Gly-OSu an optimal reagent for synthesizing compounds.
Boc-Gly-OSu (CAS 3392-07-2) References
- A COFRADIC protocol to study protein ubiquitination. | Stes, E., et al. 2014. J Proteome Res. 13: 3107-13. PMID: 24816145
- TAG-TMTpro, a Hyperplexing Quantitative Approach for High-Throughput Proteomic Studies. | Wu, Z., et al. 2022. Anal Chem. 94: 12565-12569. PMID: 36066113
- Use of thiol acids in peptide segment coupling in non-aqueous solvents. | Yamashiro, D. and Blake, J. 1981. Int J Pept Protein Res. 18: 383-92. PMID: 7309385
- Facile synthesis of amino acid and peptide esters under mild conditions via cesium salts. | Wang S-S,., et al. 1977. J Org Chem. 42: 1286-90. PMID: 845711
- Design and Synthesis of Dipeptide Nerve Growth Factor Loop 1 Mimetics and In Vitro Studies of their Neuroprotective and Differentiation-Inducing Activities | A. V. Tarasyuk, N. M. Sazonova, A. G. Rebeko, I. O. Logvinov, S. V. Nikolaev, T. A. Antipova, T. A. Gudasheva & S. B. Seredenin. (2021). Pharmaceutical Chemistry Journal. volume 54,: pages 1126–1135.
- High specific activity tritium labelling of biologically active small peptides and a related analogue | , et al. (2016). Journal of Radioanalytical and Nuclear Chemistry. volume 307,: pages 549–553.
- Synthesis of Peptides with α,β-Dehydroamino Acids, V. Coupling experiments with C-Terminal Dehydrophenylalanine and Dehydroalanine Residues | and Maciej Makowski, Barbara Rzeszotarska, Zbigniew Kubica, Grzegorz Pietrzyński, Jacek Hetper. 12. Juni 1986. Volume1986, Issue6: Pages 980-991.
- Macromolecular analogs of the copper(II) binding site of human serum albumin. 3. Synthesis, conformation, and ion binding properties of glycylglycyl-α,γ-diaminobutyric acid derivatives of poly(L-lysine) | M. T. Foffani, M. Cestaro, A. Pezzoli, and E. Peggion. 1986. Macromolecules. 19, 4,:, 945–952.
- Total synthesis of clavamine, insecticidally active compound isolated from venom of joro spider (Nephila Clavata) | T Teshima, T Matsumoto, M Miyagawa, T Wakamiya… - Tetrahedron, 1990 - Elsevier. 1990,. Tetrahedron Volume 46, Issue 11, 1990, Pages 3819-3822. Volume 46, Issue 11,: Pages 3819-3822.
- Syntheses of 3′-substituted-2-(carboxycyclopropyl)glycines via intramolecular cyclopropanation. The folded form of L-glutamate activates the Non-NMda receptor subtype | K Shimamoto, Y Ohfune - Tetrahedron letters, 1990 - Elsevier. 1990,. Tetrahedron Letters. Volume 31, Issue 28,: Pages 4049-4052.
- Preparations and Characterizations of Novel N, N′-Ethylene-Bridged-(S)-Histidyl-(S)-Tyrosine Derivatives and Their Copper(II) Complexes as Models of Galactose Oxidase | Yamato Kazuhiro 1, Inada Takanori 1, Doe Matsumi 1, Ichimura Akio 1, Takui Takeji 1, Kojima Yoshitane 1, Kikunaga Toshimitsu 2, Nakamura Shin 2, Yanagihara Naohisa 2, Onaka Tomoko 3, Yano Shigenobu 3. 2000,. Bulletin of the Chemical Society of Japan. Vol.73, No.4: 903-912.
- Pd-catalyzed one-pot chemoselective hydrogenation protocol for the preparation of carboxamides directly from azides | SN Bavikar, DB Salunke, BG Hazra, VS Pore… - Tetrahedron …, 2010 - Elsevier. 21 July 2010,. Tetrahedron Letters. Volume 51, Issue 29,: Pages 3815-3819.
- A facile synthesis of ascamycin and related analogues | J Castro-Pichel, MT García-López, G Federico - Tetrahedron, 1987 - Elsevier. Pages 383-389. Tetrahedron. Volume 43, Issue 2, 1987,: Pages 383-389.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Boc-Gly-OSu, 5 g | sc-227474 | 5 g | $68.00 |
