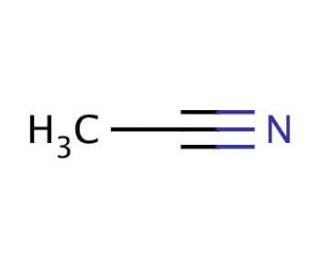
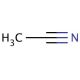
Acetonitrile (CAS 75-05-8)
Voir les citations produits (1)
ACCÈS RAPIDE AUX LIENS
L'acétonitrile, dont le numéro CAS est 75-05-8, est un liquide incolore et volatil qui est le nitrile organique le plus simple. Il est synthétisé principalement par déshydratation de l'acide acétique et de l'ammoniac ou comme sous-produit de la fabrication de l'acrylonitrile. L'acétonitrile est très prisé dans le domaine de la chimie analytique, en particulier comme solvant aprotique polaire dans la chromatographie liquide à haute performance (CLHP) et la spectrométrie de masse. Sa faible viscosité et sa force d'élution élevée en font un solvant idéal pour séparer les substances peu solubles dans l'eau, ce qui lui permet de transporter rapidement et efficacement une large gamme d'espèces chimiques à travers une colonne chromatographique. L'excellente miscibilité de ce solvant avec l'eau et d'autres solvants organiques renforce encore son utilité dans la création de divers mélanges de solvants afin d'optimiser la résolution et la détection d'analytes dans des mélanges complexes. En chimie organique de synthèse, l'acétonitrile est utilisé comme solvant pour effectuer des réactions anioniques en raison de sa capacité à dissoudre les électrolytes et de sa nature inerte dans les conditions de réaction. Il est également utilisé dans la synthèse de produits pharmaceutiques, agrochimiques et de parfums, où il sert de solvant ou d'intermédiaire. En outre, dans la recherche non biologique, les propriétés de l'acétonitrile sont exploitées dans la synthèse de l'ADN et le séquençage des peptides, facilitant la solubilisation des nucléotides et des peptides, respectivement, et améliorant le rendement et la pureté de ces biomolécules.
Acetonitrile (CAS 75-05-8) Références
- Dégradation continue de l'acétonitrile dans un bioréacteur à lit compact. | Manolov, T., et al. 2005. Appl Microbiol Biotechnol. 66: 567-74. PMID: 15630519
- Quantification du tryptophane et d'autres acides aminés plasmatiques par chromatographie liquide haute performance automatisée avec dérivatisation de l'o-phtaldialdéhyde avant la colonne: amélioration de la préparation des échantillons. | Uhe, AM., et al. 1991. J Chromatogr. 564: 81-91. PMID: 1860936
- Préparation d'un biofloculant en utilisant l'acétonitrile comme seule source d'azote et son application dans l'élimination des métaux lourds. | Fan, HC., et al. 2019. J Hazard Mater. 363: 242-247. PMID: 30308363
- Synthèse d'esters via une estérification de Steglich plus verte dans l'acétonitrile. | Lutjen, AB., et al. 2018. J Vis Exp.. PMID: 30451233
- L'acétonitrile permet de remplacer indirectement les détergents lipidiques non deutérés par des détergents lipidiques deutérés pour l'étude par résonance magnétique nucléaire des protéines solubles dans les détergents. | Wang, X., et al. 2021. Protein Sci. 30: 2324-2332. PMID: 34462977
- Modélisation du pKa des bases de Brønsted comme approche de l'énergie de Gibbs du proton dans l'acétonitrile. | Glasovac, Z. and Kovačević, B. 2022. Int J Mol Sci. 23: PMID: 36142490
- Comparaison de la capacité et de l'activité de libération d'hydrure de composés benzoheterocycliques à cinq et six membres dans l'acétonitrile. | Zhang, JY. and Zhu, XQ. 2022. Molecules. 27: PMID: 36364079
- Extraction directe à l'acétonitrile de l'huile de chanvre pour l'analyse des pesticides à l'aide de la chromatographie en phase gazeuse bidimensionnelle et de la spectrométrie de masse à triple quadripôle. | Arena, A., et al. 2023. J Chromatogr A. 1710: 464432. PMID: 37832462
- Potentialisation par l'acétone de la toxicité aiguë de l'acétonitrile chez le rat. | Freeman, JJ. and Hayes, EP. 1985. J Toxicol Environ Health. 15: 609-21. PMID: 4046068
- Transitions conformationnelles induites par l'acétonitrile dans la poly-L-lysine. | Arunkumar, AI., et al. 1997. Int J Biol Macromol. 21: 299-305. PMID: 9493053
Informations pour la commande
| Nom du produit | Ref. Catalogue | COND. | Prix HT | QTÉ | Favoris | |
Acetonitrile, 500 ml | sc-207252 | 500 ml | $57.00 | |||
Acetonitrile, 2.5 L | sc-207252A | 2.5 L | $168.00 |
