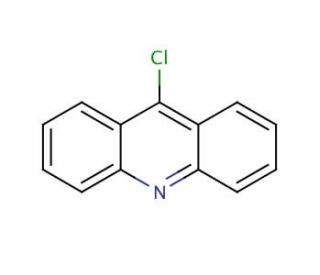
Molecular structure of 9-Chloroacridine, CAS Number: 1207-69-8
9-Chloroacridine (CAS 1207-69-8)
Application:
9-Chloroacridine is a chromogenic substrate for dapsone spectrophotometric analysis
CAS Number:
1207-69-8
Molecular Weight:
213.66
Molecular Formula:
C13H8ClN
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
9-Chloroacridine is a derivative of acridine, which contains a chlorine atom at position 9 of the acridine nucleus. It is a chlorinated acridine compound used in spectrophotometric analyses and as a reagent for the synthesis of chalcones, acridine derivatives, and quinoline derivatives. It was shown to have significant cytotoxicity in cell culture and to react with hydrochloric acid to form two chemical species: amines and protonated molecules. Molecular docking analysis has been used to show that 9-chloroacridine interacts with the enzyme dihydropteridine reductase, which is involved in pteridine synthesis.
9-Chloroacridine (CAS 1207-69-8) References
- Complexes of acridine and 9-chloroacridine with I2: formation of unusual I6 chains through charge-transfer interactions involving amphoteric I2. | Rimmer, EL., et al. 2000. Chemistry. 6: 4071-81. PMID: 11128273
- Colorimetric determination of isoniazid with 9-chloroacridine. | Stewart, JT. and Settle, DA. 1975. J Pharm Sci. 64: 1403-5. PMID: 1151717
- Synthesis and anti-inflammatory evaluation of 9-phenoxyacridine and 4-phenoxyfuro[2,3-b]quinoline derivatives. Part 2. | Chen, YL., et al. 2003. Bioorg Med Chem. 11: 3921-7. PMID: 12927852
- Arylethynylacridines: electrochemiluminescence and photophysical properties. | Elangovan, A., et al. 2004. Org Biomol Chem. 2: 3113-8. PMID: 15505716
- Synthesis and antitumor activity of 5-(9-acridinylamino)anisidine derivatives. | Bacherikov, VA., et al. 2005. Bioorg Med Chem. 13: 6513-20. PMID: 16140018
- Synthesis of new chalcone derivatives containing acridinyl moiety with potential antimalarial activity. | Tomar, V., et al. 2010. Eur J Med Chem. 45: 745-51. PMID: 20022412
- Spectrophotometric determination of dapsone by using 9-chloroacridine as a chromogenic reagent. | Shoukrallah, I., et al. 1990. Pharmazie. 45: 675-7. PMID: 2284310
- A magnetic nanoparticle-supported N-heterocyclic carbene-palladacycle: an efficient and recyclable solid molecular catalyst for Suzuki-Miyaura cross-coupling of 9-chloroacridine. | Deng, Q., et al. 2017. Chem Commun (Camb). 53: 13063-13066. PMID: 29165443
- An electrochemical study of 9-chloroacridine redox behavior and its interaction with double-stranded DNA. | Rupar, J., et al. 2020. Bioelectrochemistry. 135: 107579. PMID: 32534381
- Can Zeolite-Supporting Acridines Boost Their Anticancer Performance? | Ranković, M., et al. 2023. J Funct Biomater. 14: PMID: 36976097
- Determination of sulfonamides and local anesthetics with 9=chloroacridine by quenching fluorometry. | Stewart, JT. and Wilkin, RE. 1972. J Pharm Sci. 61: 432-3. PMID: 4259033
- Colorimetric determination of hydralazine with 9-chloroacridine. | Stewart, JT. and Chang, YC. 1979. J Assoc Off Anal Chem. 62: 1107-9. PMID: 528456
- Colorimetric determination of some sulfonamides with 9-chloroacridine. | Stewart, JT., et al. 1969. J Pharm Sci. 58: 1261-2. PMID: 5349116
- Improved colorimetric determination of primary aromatic amines with 9-chloroacridine: application to some local anesthetics. | Stewart, JT. and Lotti, DM. 1970. J Pharm Sci. 59: 838-40. PMID: 5423089
Substrate of:
Chromogenic.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
9-Chloroacridine, 1 g | sc-227140 | 1 g | $63.00 |
