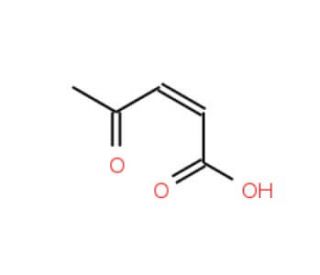
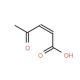
3-Acetylacrylic acid (CAS 4743-82-2)
QUICK LINKS
3-Acetylacrylic acid, also known as 3-AAA, is an innovative compound derived from the metabolic processes of select plant species. Its natural origin and versatile properties have led to its extensive utilization in the fields of biochemistry and physiology. This substance finds applications in various laboratory experiments due to its remarkable characteristics. A 3-Acetylacrylic acid is a tool for studying metabolism and quantifying the rate of metabolic processes. One of the key attributes of 3-Acetylacrylic acid is its unique mechanism of action, which contributes to its wide applicability in scientific research. It functions as an inhibitor of acetyl-CoA carboxylase, an enzyme involved in fatty acid synthesis. By inhibiting this enzyme, 3-Acetylacrylic acid effectively reduces the production of fatty acids, leading to diverse effects on the body. Furthermore, 3-Acetylacrylic acid acts as an inhibitor of 3-hydroxybutyryl-CoA dehydrogenase, an enzyme involved in fatty acid metabolism.
3-Acetylacrylic acid (CAS 4743-82-2) References
- Antistaphylococcic factors in brain extract. | BERGER, AJ., et al. 1957. Nature. 179: 588-9. PMID: 13418740
- A probable peptidase in carcinoid tumours. | PEARSE, AG. and PEPLER, WJ. 1957. Nature. 179: 589. PMID: 13418741
- Polymerization of nonfood biomass-derived monomers to sustainable polymers. | Zhang, Y. and Chen, EY. 2014. Top Curr Chem. 353: 185-227. PMID: 24699900
- Liquid phase oxidation chemistry in continuous-flow microreactors. | Gemoets, HP., et al. 2016. Chem Soc Rev. 45: 83-117. PMID: 26203551
- Study on decomposition products of norbixin during bleaching with hydrogen peroxide and a peroxidase by means of UPLC-UV and mass spectrometry. | van Scheppingen, WB., et al. 2012. Food Chem. 132: 1354-1359. PMID: 29243622
- An electrocatalytic route for transformation of biomass-derived furfural into 5-hydroxy-2(5H)-furanone. | Wu, H., et al. 2019. Chem Sci. 10: 4692-4698. PMID: 31123580
- Poly(4-ketovalerolactone) from Levulinic acid: Synthesis and Hydrolytic Degradation. | Xu, S., et al. 2020. Macromolecules. 53: 4952-4959. PMID: 33767514
- Degradation of dioxin-like compounds by microorganisms. | Wittich, RM. 1998. Appl Microbiol Biotechnol. 49: 489-99. PMID: 9650248
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Acetylacrylic acid, 1 g | sc-275950 | 1 g | $520.00 |
