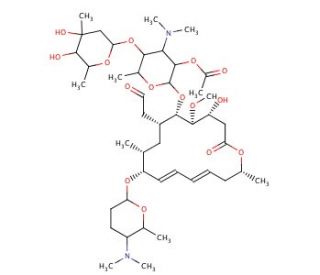
2′-O-Acetylspiramycin I (CAS 89000-32-8)
QUICK LINKS
2′-O-Acetylspiramycin I is a modified derivative of spiramycin, a macrolide antibiotic, which is utilized in scientific research to explore the chemical and biochemical properties of macrolide structures. This specific modification, the acetylation at the 2′-O position of spiramycin, impacts the molecular interactions and stability of the compound, making it a valuable tool for studying the effects of structural changes on the activity and properties of macrolide antibiotics. In research contexts, 2′-O-Acetylspiramycin I is particularly useful for investigating the mechanisms of antibiotic resistance and the binding efficiency of macrolides to their ribosomal targets. The introduction of the acetyl group can affect the compound′s binding affinity to bacterial ribosomes, providing insights into the nuances of drug-target interactions. This modification also offers an avenue to study how changes in molecular structure can influence the solubility and permeability of macrolides, critical factors in the development of new antibiotics. Furthermore, 2′-O-Acetylspiramycin I serves as a model compound in synthetic organic chemistry for developing new synthetic strategies and methodologies. Researchers utilize this compound to refine techniques in acetylation reactions, enhance understanding of the steric and electronic effects of acetyl substitutions, and develop novel derivatives with potentially improved properties. This compound′s role in research helps in advancing our understanding of macrolide antibiotics′ chemistry and their interaction dynamics.
2′-O-Acetylspiramycin I (CAS 89000-32-8) References
- 4'''-N-demethylspiramycin derivatives: synthesis and evaluation of effectiveness against drug-resistant bacteria. | Sunazuka, T., et al. 2008. J Antibiot (Tokyo). 61: 175-84. PMID: 18503196
- Chemical modification of spiramycins. III. Synthesis and antibacterial activities of 4''-sulfonates and 4''-alkylethers of spiramycin I. | Sano, H., et al. 1984. J Antibiot (Tokyo). 37: 750-9. PMID: 6469869
- Chemical modification of spiramycins. I. Synthesis of the acetal derivatives of neospiramycin I. | Sano, H., et al. 1983. J Antibiot (Tokyo). 36: 1336-44. PMID: 6643282
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2'-O-Acetylspiramycin I, 10 mg | sc-206551 | 10 mg | $330.00 |
