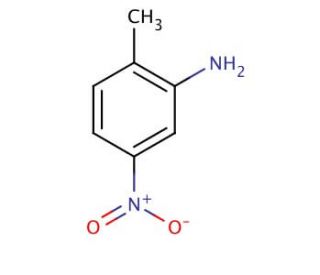
2-Methyl-5-nitroaniline (CAS 99-55-8)
Direktverknüpfungen
2-Methyl-5-Nitroanilin, oder 2-MNA, ist eine bemerkenswerte aromatische Aminkomponente, die weitläufig in verschiedenen wissenschaftlichen Anwendungen verwendet wird, dank seiner einzigartigen Eigenschaften. Es präsentiert sich als ein blassgelbes, kristallines Feststoff und besitzt einen Schmelzpunkt von ca. 100°C. Die Vielseitigkeit des Stoffes ist offensichtlich, da er sowohl in Wasser als auch in organischen Lösungsmitteln löslich ist und im kommerziellen Markt in flüssiger und fester Form leicht erhältlich ist. Neben seiner kommerziellen Verfügbarkeit ist seine chemische Struktur relativ einfach, was ihn zu einer relativ stabilen Substanz macht. Seine Bedeutung in der wissenschaftlichen Forschung ist enorm, wo er als Reagenz in der organischen Synthese, als Katalysator für chemische Reaktionen und als Reaktans in der Synthese verschiedener Verbindungen eingesetzt wird. Darüber hinaus erstreckt sich die Anwendung des Stoffes auf die Synthese von Agrochemikalien und verschiedenen anderen Materialien, was weiterhin seine Bedeutung für wissenschaftliche Fortschritte hervorhebt. Die Wirkungsweise von 2-Methyl-5-Nitroanilin liegt in seiner Fähigkeit, mit anderen Molekülen kovalente Bindungen einzugehen, aufgrund seiner aromatischen Aminkomponente. Abhängig vom Umfeld kann es sowohl als Elektronendonator als auch als Akzeptor wirken. Darüber hinaus kommen seine katalytischen Eigenschaften zum Einsatz, um den Ablauf chemischer Reaktionen effektiv zu beschleunigen. Die Vielseitigkeit und die einzigartigen Eigenschaften von 2-Methyl-5-Nitroanilin machen es zu einer unverzichtbaren Verbindung, die erheblich zu verschiedenen wissenschaftlichen Unternehmungen und Anwendungen in verschiedenen Bereichen beiträgt.
2-Methyl-5-nitroaniline (CAS 99-55-8) Literaturhinweise
- Synthese und antikonvulsive und neurotoxische Eigenschaften von substituierten N-Phenylderivaten des Phthalimid-Pharmakophores. | Vamecq, J., et al. 2000. J Med Chem. 43: 1311-9. PMID: 10753468
- Experimenteller Beweis für die Nicht-Planarität der Aminogruppe in Nitroanilinen: Neutronenbeugungsstudie von 2-Methyl-5-nitroanilin bei 100 K. | Ellena, J., et al. 1999. Acta Crystallogr B. 55: 209-215. PMID: 10927358
- 4-Iodo-2-methyl-5-nitroanilin zeigt weder starke Wasserstoffbrückenbindungen noch intermolekulare I..nitro-Wechselwirkungen. | Garden, SJ., et al. 2001. Acta Crystallogr C. 57: 1212-4. PMID: 11600789
- Tensid-unterstützte UV-Photolyse von Nitroarenen. | Diehl, CA., et al. 2002. Chemosphere. 46: 553-60. PMID: 11838434
- Neubewertung der Verstärkung großer Dipolmomente in Kristallen: eine detaillierte experimentelle und theoretische Ladungsdichteanalyse von 2-Methyl-4-nitroanilin. | Whitten, AE., et al. 2006. J Phys Chem A. 110: 8763-76. PMID: 16836439
- Etablierung einer Extraktionsmethode für die Rückgewinnung von Tätowierungspigmenten aus menschlicher Haut unter Verwendung der HPLC-Diodenarray-Detektor-Technologie. | Engel, E., et al. 2006. Anal Chem. 78: 6440-7. PMID: 16970319
- Eine neue Klasse von funktionalisierten Polyoxometalaten: Synthese, Struktur und vorläufige Studien zur Antitumoraktivität von drei Arylimido-substituierten Hexamolybdaten, die eine stark elektronenziehende Nitrogruppe tragen, (Bu4N)2[Mo6O18([Dreifachbindung]NAr)] (Ar = 3-NO2-C6H4, 2-CH3-4-NO2-C6H3, 2-CH3-5-NO2-C6H3). | Xue, S., et al. 2008. Dalton Trans. 4770-5. PMID: 18728886
- Über die Topologie der Elektronendichte und die elektrostatischen Eigenschaften von Nitroanilinen. Eine theoretische Untersuchung der Kristalle von m-Nitroanilin und 2-Methyl-5-Nitroanilin. | Fantoni, AC., et al. 2009. J Phys Chem A. 113: 9527-32. PMID: 19653659
- Das Tätowieren der Haut führt zum Transport und zur lichtinduzierten Zersetzung von Tätowierpigmenten - eine erste Quantifizierung in vivo anhand eines Mausmodells. | Engel, E., et al. 2010. Exp Dermatol. 19: 54-60. PMID: 19703227
- Ein neues Polymorph von 2-Methyl-6-Nitroanilin. | Callear, SK. and Hursthouse, MB. 2009. Acta Crystallogr C. 65: o539-42. PMID: 19805890
- Röntgenbeugung, Schwingungs- und quantenchemische Untersuchungen von 2-Methyl-4-nitroanilinium-Trichloracetat-Trichloressigsäure. | Arjunan, V., et al. 2012. Spectrochim Acta A Mol Biomol Spectrosc. 97: 625-38. PMID: 22858610
- Imatinib-Zwischenprodukt als Zwei-in-Eins-Zweikanal-Sensor für die Erkennung von Cu²⁺- und I-Ionen in wässrigen Medien und seine praktischen Anwendungen. | Patil, SR., et al. 2014. Dalton Trans. 43: 13299-306. PMID: 25056090
- Die Dissoziationskonstanten von einigen disubstituierten Anilinen und Phenolen in wässriger Lösung bei 25° C. | Robinson, RA. 1967. J Res Natl Bur Stand A Phys Chem. 71A: 213-218. PMID: 31824047
- Strukturelle, theoretische und spektroskopische Analyse von 2-Methyl-5-Nitroanilin-Salzen mit verschiedenen anorganischen Säuren. | Medviediev, V. and Daszkiewicz, M. 2019. Acta Crystallogr B Struct Sci Cryst Eng Mater. 75: 1003-1013. PMID: 32830680
- Bewertung von Kohlenstoff-11-markiertem 5-(1-Methyl-1H-pyrazol-4-yl)-N-(2-Methyl-5-(3-(trifluormethyl)benzamido)phenyl)nikotinamid als PET-Tracer zur Darstellung der CSF-1R-Expression im Gehirn. | van der Wildt, B., et al. 2021. Bioorg Med Chem. 42: 116245. PMID: 34119698
Bestellinformation
| Produkt | Katalog # | EINHEIT | Preis | ANZAHL | Favoriten | |
2-Methyl-5-nitroaniline, 100 g | sc-238140 | 100 g | $23.00 |
