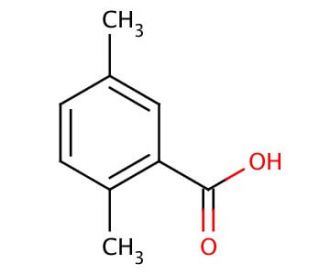
2,5-Dimethylbenzoic acid (CAS 610-72-0)
QUICK LINKS
2,5-Dimethylbenzoic acid is a crystalline solid of white color, displaying limited solubility in water but dissolving readily in prevalent organic solvents like ethanol, methanol, acetone, and benzene. It is used as a foundational component in synthesizing a range of organic compounds, including agrochemicals, polymers, and dyes. 2,5-Dimethylbenzoic acid serves as a standard for numerous analytical methods such as HPLC, GC, and MS. It may act to have a functional role in the production of proteins that are involved in cellular signaling pathways such as chemotaxis and apoptosis. 2,5-Dimethylbenzoic acid can function as a catalyst in reactions involving various organic compounds, act as a ligand binding to diverse organic molecules, and serve as an inhibitor in reactions with several organic molecules. 2,5-Dimethylbenzoic acid is a metabolite of pseudocumene (1,2,4-trimethylbenzene).
2,5-Dimethylbenzoic acid (CAS 610-72-0) References
- Toxicokinetics and metabolism of pseudocumene (1,2,4-trimethylbenzene) after inhalation exposure in rats. | Swiercz, R., et al. 2002. Int J Occup Med Environ Health. 15: 37-42. PMID: 12038862
- Oxidation of naphthenoaromatic and methyl-substituted aromatic compounds by naphthalene 1,2-dioxygenase. | Selifonov, SA., et al. 1996. Appl Environ Microbiol. 62: 507-14. PMID: 16535238
- Precursor-Directed Combinatorial Biosynthesis of Cinnamoyl, Dihydrocinnamoyl, and Benzoyl Anthranilates in Saccharomyces cerevisiae. | Eudes, A., et al. 2015. PLoS One. 10: e0138972. PMID: 26430899
- A Facile Solid-Phase Route to Renewable Aromatic Chemicals from Biobased Furanics. | Thiyagarajan, S., et al. 2016. Angew Chem Int Ed Engl. 55: 1368-71. PMID: 26684008
- Selectivity Control in the Tandem Aromatization of Bio-Based Furanics Catalyzed by Solid Acids and Palladium. | Genuino, HC., et al. 2017. ChemSusChem. 10: 277-286. PMID: 27557889
- Superelectrophiles in Synthesis: Preparation of Aromatic Imides. | Kennedy, SH., et al. 2019. J Org Chem. 84: 14133-14140. PMID: 31571485
- Maydisone, a novel oxime polyketide from the cultures of Bipolaris maydis. | Tran, TM., et al. 2022. Nat Prod Res. 36: 102-107. PMID: 32400180
- Intermediates formed during natural attenuation of C9 aromatics under simulated marine conditions: Identification, transformation pathway, and toxicity to microalgae. | Zhang, B., et al. 2022. Environ Res. 206: 112558. PMID: 34932976
- The Interplay between Kinetics and Thermodynamics in Furan Diels-Alder Chemistry for Sustainable Chemicals Production. | Cioc, RC., et al. 2022. Angew Chem Int Ed Engl. 61: e202114720. PMID: 35014138
- Bioactive Components and Health Potential of Endophytic Micro-Fungal Diversity in Medicinal Plants. | Muthukrishnan, S., et al. 2022. Antibiotics (Basel). 11: PMID: 36358188
- Biosynthetic studies on aromatic carotenoids. Biosynthesis of chlorobactene. | Moshier, SE. and Chapman, DJ. 1973. Biochem J. 136: 395-404. PMID: 4774401
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,5-Dimethylbenzoic acid, 10 g | sc-231127 | 10 g | $52.00 |
