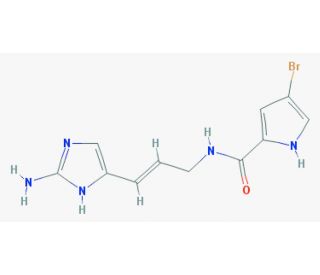
Hymenidin (CAS 107019-95-4)
QUICK LINKS
Hymenidin, an alkaloid extracted from marine sponges of the genus Agelas, is widely researched for its bioactive properties in cell signaling and communication pathways. This compound is particularly notable for its ability to modulate ion channels, specifically targeting sodium and calcium channels, which are crucial for the propagation of electrical signals in cells. By influencing these channels, hymenidin offers a unique tool for studying the mechanisms of nerve impulse transmission and muscle contraction. Additionally, its role in affecting cell communication extends to research on cellular defense mechanisms, where hymenidin′s ability to disrupt microbial communication—known as quorum sensing—provides insights into microbial ecology and the potential for controlling biofilm formation. This compound′s diverse biological activities make it a subject of interest for understanding the complex interactions within marine ecosystems and the evolutionary adaptations of marine organisms to their environments. Research utilizing hymenidin thus contributes significantly to the fields of neurobiology, pharmacology, and marine biology, providing broader implications for the study of natural compounds in regulating biological processes and maintaining ecological balance.
Hymenidin (CAS 107019-95-4) References
- Inhibition of cyclin-dependent kinases, GSK-3beta and CK1 by hymenialdisine, a marine sponge constituent. | Meijer, L., et al. 2000. Chem Biol. 7: 51-63. PMID: 10662688
- Sventrin, a new bromopyrrole alkaloid from the Caribbean sponge Agelas sventres. | Assmann, M., et al. 2001. J Nat Prod. 64: 1593-5. PMID: 11754625
- Brominated pyrrole alkaloids from marine Agelas sponges reduce depolarization-induced cellular calcium elevation. | Bickmeyer, U., et al. 2004. Toxicon. 44: 45-51. PMID: 15225561
- Antineoplastic agents 470. Absolute configuration of the marine sponge bromopyrrole agelastatin A. | Pettit, GR., et al. 2005. Oncol Res. 15: 11-20. PMID: 15839302
- (-)-Agelasidine A from Agelas clathrodes. | Medeiros, MA., et al. 2006. Z Naturforsch C J Biosci. 61: 472-6. PMID: 16989304
- Total syntheses of oroidin, hymenidin and clathrodin. | Rasapalli, S., et al. 2013. Org Biomol Chem. 11: 4133-7. PMID: 23695419
- The marine sponge Agelas citrina as a source of the new pyrrole-imidazole alkaloids citrinamines A-D and N-methylagelongine. | Cychon, C., et al. 2015. Beilstein J Org Chem. 11: 2029-37. PMID: 26664624
- Integrated discovery of FOXO1-DNA stabilizers from marine natural products to restore chemosensitivity to anti-EGFR-based therapy for metastatic lung cancer. | Sun, Y., et al. 2017. Mol Biosyst. 13: 330-337. PMID: 27966721
- Clathrodin, hymenidin and oroidin, and their synthetic analogues as inhibitors of the voltage-gated potassium channels. | Zidar, N., et al. 2017. Eur J Med Chem. 139: 232-241. PMID: 28802123
- Bromopyrrole Alkaloid Inhibitors of the Proteasome Isolated from a Dictyonella sp. Marine Sponge Collected at the Amazon River Mouth. | de Souza, RTMP., et al. 2018. J Nat Prod. 81: 2296-2300. PMID: 30281303
- Total Synthesis of (±)-Sceptrin. | Nguyen, LV. and Jamison, TF. 2020. Org Lett. 22: 6698-6702. PMID: 32379973
- Feature-Based Molecular Networking Discovery of Bromopyrrole Alkaloids from the Marine Sponge Agelas dispar. | Freire, VF., et al. 2022. J Nat Prod. 85: 1340-1350. PMID: 35427139
- A novel antagonist of serotonergic receptors, hymenidin, isolated from the Okinawan marine sponge Hymeniacidon sp. | Kobayashi, J., et al. 1986. Experientia. 42: 1176-7. PMID: 3770140
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hymenidin, 1 mg | sc-202177 | 1 mg | $263.00 |
