Uramil (CAS 118-78-5)
QUICK LINKS
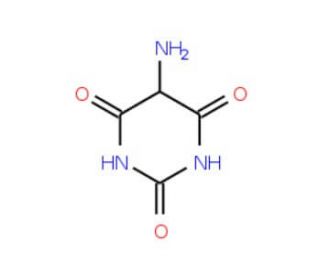
Uramil, a heterocyclic compound, emerges from the methylation of uracil, leading to thymine, a key player in DNA replication. This compound, with its yellowish-white hue, easily dissolves in water and ethanol. Its cytotoxic nature arises from its diverse functional groups, such as benzyl, alkylthio, hydroxyl, and carbonyl. Upon reacting with cellular nitrogen atoms, Uramil instigates a redox cycle that gives birth to reactive oxygen species. The compound′s glucose analogue not only enhances its solubility but also simplifies its purification process. In the realm of scientific research, Uramil finds its place in tasks like DNA and RNA synthesis, protein stabilization, and enzyme conservation. Moreover, it′s a foundation stone for crafting various pyrimidine derivatives and is instrumental in gene knockout studies.
Uramil (CAS 118-78-5) References
- Relation between triketone structure, generation of reactive oxygen species, and selective toxicity of the diabetogenic agent alloxan. | Elsner, M., et al. 2008. Antioxid Redox Signal. 10: 691-9. PMID: 18177230
- Structural requirements of alloxan and ninhydrin for glucokinase inhibition and of glucose for protection against inhibition. | Lenzen, S., et al. 1988. Br J Pharmacol. 95: 851-9. PMID: 3207996
- Oxidation of uric acid. 4. Synthesis, structure, and diabetogenic action of 5-imino-2,4,6(1H,3H,5H)-pyrimidinetrione salts and their alloxan-like covalent adducts. | Poje, M., et al. 1983. J Med Chem. 26: 861-4. PMID: 6854588
- Indirect electrochemical oxidation of benzylamine using 5-amino-6-hydroxy-2,4(1H, 3H)-pyrimidinedione as a current mediator | Sotaro Ito, Kazuo Sasaki, Yasuo Yanagi. 1982. Bioelectrochemistry and Bioenergetics. 9: 313-324.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Uramil, 1 g | sc-296683 | 1 g | $88.00 | |||
Uramil, 5 g | sc-296683A | 5 g | $327.00 |
