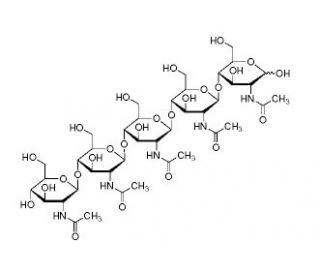
Penta-N-acetylchitopentaose (CAS 36467-68-2)
QUICK LINKS
Penta-N-acetylchitopentaose is a complex carbohydrate compound derived from chitin, a naturally occurring polysaccharide found in the exoskeletons of crustaceans and the cell walls of fungi. It consists of five N-acetylglucosamine (GlcNAc) units linked together by β-1,4-glycosidic bonds. Penta-N-acetylchitopentaose is characterized by its repeating GlcNAc units, each of which is acetylated at the amino group. The acetyl groups impart stability and solubility to the molecule, making it suitable for various biochemical and biotechnological applications. This chemical compound is widely used in the field of glycobiology for studying carbohydrate-protein interactions and enzymatic reactions. Penta-N-acetylchitopentaose serves as a valuable substrate for enzymes such as chitinases and chitin-binding proteins, allowing researchers to investigate their specificity, catalytic mechanisms, and structural characteristics. In addition, Penta-N-acetylchitopentaose has been employed in drug delivery systems and as a precursor for the synthesis of other chitin derivatives. Its ability to self-assemble into nanoparticles or hydrogels has attracted interest in the development of nanocarriers for targeted drug delivery or tissue engineering applications. Moreover, Penta-N-acetylchitopentaose plays a role in understanding the biological functions of chitin and chitin-binding proteins in various organisms. By studying its interactions with lectins and other carbohydrate-binding proteins, researchers can elucidate the recognition processes involved in host-pathogen interactions, immune responses, and cell signaling pathways. Overall, Penta-N-acetylchitopentaose is useful in glycobiology and related fields due to its structural complexity, solubility, and versatility. Its applications extend to enzyme kinetics studies, drug delivery systems, and investigations of carbohydrate-protein interactions, contributing to our understanding of the roles carbohydrates play in biological processes.
Penta-N-acetylchitopentaose (CAS 36467-68-2) References
- Mode of action of chitin deacetylase from Mucor rouxii on N-acetylchitooligosaccharides. | Tsigos, I., et al. 1999. Eur J Biochem. 261: 698-705. PMID: 10215886
- Chitinous materials inhibit nitric oxide production by activated RAW 264.7 macrophages. | Hwang, SM., et al. 2000. Biochem Biophys Res Commun. 271: 229-33. PMID: 10777707
- Induced accumulation of 2-hydroxy-4,7-dimethoxy-1,4-benzoxazin-3-one glucoside (HDMBOA-Glc) in maize leaves. | Oikawa, A., et al. 2001. Phytochemistry. 56: 669-75. PMID: 11314951
- Induction of anthranilate synthase activity by elicitors in oats. | Matsukawa, T., et al. 2002. Z Naturforsch C J Biosci. 57: 121-8. PMID: 11926523
- A novel beta-N-acetylglucosaminidase of Clostridium paraputrificum M-21 with high activity on chitobiose. | Li, H., et al. 2002. Appl Microbiol Biotechnol. 60: 420-7. PMID: 12466882
- [Biosynthesis of chitooligosaccharides by recombinant Escherichia coli]. | Zhang, DW., et al. 2007. Sheng Wu Gong Cheng Xue Bao. 23: 525-9. PMID: 17578006
- A two-step fermentation process for efficient production of penta-N-acetyl-chitopentaose in recombinant Escherichia coli. | Zhang, D., et al. 2007. Biotechnol Lett. 29: 1729-33. PMID: 17710376
- Concise synthesis of 4-methylumbelliferyl-penta-N-acetylchitopentaoside and its inhibition effect on chitinase. | Huang, GL. 2009. Int J Biol Macromol. 45: 381-3. PMID: 19635498
- Experimental Measurements and Mathematical Modeling of Cytosolic Ca(2+) Signatures upon Elicitation by Penta-N-acetylchitopentaose Oligosaccharides in Nicotiana tabacum Cell Cultures. | Mrozek, K., et al. 2013. Plants (Basel). 2: 750-68. PMID: 27137402
- Structural and biochemical insights into an insect gut-specific chitinase with antifungal activity. | Liu, T., et al. 2020. Insect Biochem Mol Biol. 119: 103326. PMID: 31968227
- AFM dendritips functionalized with molecular probes specific to cell wall polysaccharides as a tool to investigate cell surface structure and organization. | Schiavone, M., et al. 2019. Cell Surf. 5: 100027. PMID: 32743143
- Isolation, macromolecular properties, and combining site of a chito-oligosaccharide-specific lectin from the exudate of ridge gourd (Luffa acutangula). | Anantharam, V., et al. 1986. J Biol Chem. 261: 14621-7. PMID: 3771544
- Transglycosylation in a two-phase aqueous-organic system with catalysis by a lipid-coated beta-D-galactosidase. | Mori, T., et al. 1997. Carbohydr Res. 298: 65-73. PMID: 9076932
- Rearrangement of actin microfilaments in plant root hairs responding to rhizobium etli nodulation signals. | Crdenas, L., et al. 1998. Plant Physiol. 116: 871-7. PMID: 9501120
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Penta-N-acetylchitopentaose, 10 mg | sc-222146 | 10 mg | $408.00 |
