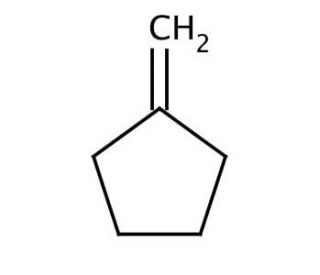
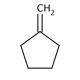
Molecular structure of Methylenecyclopentane, CAS Number: 1528-30-9
Methylenecyclopentane (CAS 1528-30-9)
CAS Number:
1528-30-9
Molecular Weight:
82.14
Molecular Formula:
C6H10
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Methylenecyclopentane (MCP) is a cyclic hydrocarbon molecule used as a starting material for the synthesis of other compounds. Also, it acts as an antioxidant by scavenging free radicals and reduces inflammation by inhibiting the production of pro-inflammatory molecules. In addition, it may act to reduce the levels of certain hormones, including cortisol and testosterone. Methylenecyclopentane has been studied for its potential use in the synthesis of polymers, the production of biofuels, and as a fuel additive.
Methylenecyclopentane (CAS 1528-30-9) References
- Ultrafast dynamics of cyclohexene and cyclohexene-d10 excited at 200 nm. | Fuss, W., et al. 2001. J Am Chem Soc. 123: 7101-8. PMID: 11459489
- New Cobalt-Catalyzed Cycloisomerization of epsilon-Acetylenic beta-Keto Esters. Application to a Powerful Cyclization Reactions Cascade. | Cruciani, P., et al. 1996. J Org Chem. 61: 2699-2708. PMID: 11667101
- Cobalt-catalyzed intramolecular Heck-type reaction of 6-halo-1-hexene derivatives. | Fujioka, T., et al. 2002. Org Lett. 4: 2257-9. PMID: 12074681
- Palladium(II) acetate mediated oxidative cyclization of omega-unsaturated alpha-cyano ketones. A facile methylenecyclopentane annulation process. | Kung, LR., et al. 2003. Chem Commun (Camb). 2490-1. PMID: 14587746
- Mechanistic investigations of the ethylene tetramerisation reaction. | Overett, MJ., et al. 2005. J Am Chem Soc. 127: 10723-30. PMID: 16045361
- Coordination copolymerization of severely encumbered isoalkenes with ethylene: enhanced enchainment mediated by binuclear catalysts and cocatalysts. | Li, H., et al. 2005. J Am Chem Soc. 127: 14756-68. PMID: 16231930
- LC-NMR and LC-MS identification of an impurity in a novel antifungal drug icofungipen. | Novak, P., et al. 2009. J Pharm Biomed Anal. 50: 68-72. PMID: 19410412
- Conia-ene annulation of the α-cyano β-TMS-capped alkynyl cycloalkanone system and its synthetic application. | Chin, CL., et al. 2011. Org Biomol Chem. 9: 4778-81. PMID: 21629924
- Development of an asymmetric trimethylenemethane cycloaddition reaction: application in the enantioselective synthesis of highly substituted carbocycles. | Trost, BM., et al. 2011. J Am Chem Soc. 133: 19483-97. PMID: 21936576
- Enantiospecific intramolecular Heck reactions of secondary benzylic ethers. | Harris, MR., et al. 2014. J Am Chem Soc. 136: 7825-8. PMID: 24852707
- Diastereoselective Synthesis of 6″-(Z)- and 6″-(E)-Fluoro Analogues of Anti-hepatitis B Virus Agent Entecavir and Its Evaluation of the Activity and Toxicity Profile of the Diastereomers. | Kumamoto, H., et al. 2016. J Org Chem. 81: 2827-36. PMID: 27009432
- Synthesis of functionalized cyclopentenes through allenic ketone-based multicomponent reactions. | Wang, Q., et al. 2018. Org Biomol Chem. 16: 8854-8858. PMID: 30411769
- NHC/Nickel(II)-Catalyzed [3+2] Cross-Dimerization of Unactivated Olefins and Methylenecyclopropanes. | Huang, JQ. and Ho, CY. 2020. Angew Chem Int Ed Engl. 59: 5288-5292. PMID: 31943646
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methylenecyclopentane, 1 g | sc-257799 | 1 g | $124.00 |
