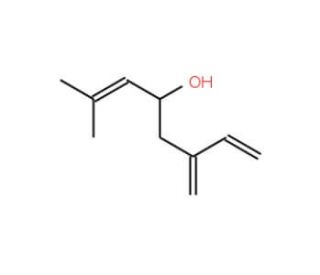
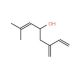
Molecular structure of Ipsdienol, CAS Number: 14434-41-4
Ipsdienol (CAS 14434-41-4)
Alternate Names:
Ipsdienol is also known as 2-Methyl-6-methyleneocta-2,7-dien-4-ol.
Application:
Ipsdienol is an insect pheromone that can attract Southern Pine Engravers and Associated Bark Beetles.
CAS Number:
14434-41-4
Purity:
≥90%
Molecular Weight:
152.236
Molecular Formula:
C10H16O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Ipsdienol is an insect pheromone. Both enantiomers of Ipsdienol can be produced in high purity and in preparative scale through the chiral resolution of its racemic precursor. It was found that Southern Pine Engravers and Associated Bark Beetles were attracted to Ipsdienol in the Southeastern United States. Ipsdienol was also demonstrated to attract Monochamus titillator and Associated Large Pine Woodborers, again in the Southeastern United States. A pheromone combination of Ipsdienol, Ianierone, and Ipsenol may be a cost-effective general lure for I. avulsus, I. grandicollis, and I. pini.
Ipsdienol (CAS 14434-41-4) References
- De novo biosynthesis of the aggregation pheromone components ipsenol and ipsdienol by the pine bark beetles Ips paraconfusus Lanier and Ips pini (Say) (Coleoptera: Scolytidae). | Seybold, SJ., et al. 1995. Proc Natl Acad Sci U S A. 92: 8393-7. PMID: 11607576
- Biochemistry and molecular biology of de novo isoprenoid pheromone production in the Scolytidae. | Seybold, SJ. and Tittiger, C. 2003. Annu Rev Entomol. 48: 425-53. PMID: 12460936
- Ipsenol and ipsdienol attract Monochamus titillator (Coleoptera: Cerambycidae) and associated large pine woodborers in southeastern United States. | Miller, DR. and Asaro, C. 2005. J Econ Entomol. 98: 2033-40. PMID: 16539130
- Functional expression of a bark beetle cytochrome P450 that hydroxylates myrcene to ipsdienol. | Sandstrom, P., et al. 2006. Insect Biochem Mol Biol. 36: 835-45. PMID: 17046597
- Myrcene hydroxylases do not determine enantiomeric composition of pheromonal ipsdienol in Ips spp. | Sandstrom, P., et al. 2008. J Chem Ecol. 34: 1584-92. PMID: 19034575
- Pheromone production in bark beetles. | Blomquist, GJ., et al. 2010. Insect Biochem Mol Biol. 40: 699-712. PMID: 20727970
- Role of ipsdienol, ipsenol, and cis-verbenol in chemical ecology of Ips avulsus, Ips calligraphus, and Ips grandicollis (Coleoptera: Curculionidae: Scolytinae). | Allison, JD., et al. 2012. J Econ Entomol. 105: 923-9. PMID: 22812131
- Enantiomeric composition of ipsdienol: A chemotaxonomic character for north American populations ofIps spp. in thepini subgeneric group (coleoptera: Scolytidae). | Seybold, SJ., et al. 1995. J Chem Ecol. 21: 995-1016. PMID: 24234415
- Response ofDendroctonus brevicomis Le Conte (Coleoptera: Scolytidae) to different release rates and ratios of aggregation semiochemicals and the inhibitors verbenone and ipsdienol. | Bertram, SL. and Paine, TD. 1994. J Chem Ecol. 20: 2931-41. PMID: 24241925
- Inter- and intrapopulation variation of the pheromone, ipsdienol produced by male pine engravers,Ips pini (Say) (Coleoptera: Scolytidae). | Miller, DR., et al. 1989. J Chem Ecol. 15: 233-47. PMID: 24271438
- Sex-specific production of ipsdienol and myrcenol byDendroctonus ponderosae (Coleoptera: Scolytidae) exposed to myrcene vapors. | Hunt, DW., et al. 1986. J Chem Ecol. 12: 1579-86. PMID: 24305835
- Ipsenol, Ipsdienol, Ethanol, and α-Pinene: Trap Lure Blend for Cerambycidae and Buprestidae (Coleoptera) in Pine Forests of Eastern North America. | Miller, DR., et al. 2015. J Econ Entomol. 108: 1837-51. PMID: 26470326
- Cytochromes P450: terpene detoxification and pheromone production in bark beetles. | Blomquist, GJ., et al. 2021. Curr Opin Insect Sci. 43: 97-102. PMID: 33359166
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ipsdienol, 1 g | sc-295190 | 1 g | $474.00 |
