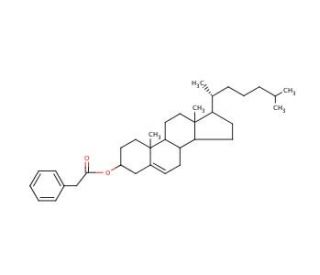
Cholesteryl phenylacetate (CAS 33998-26-4)
QUICK LINKS
Cholesteryl phenylacetate, a cholesteryl ester compound, is distinguished by its integration of a cholesterol moiety with a phenylacetate group. This molecular architecture imparts distinct physicochemical properties that make it a valuable compound in various research domains, particularly in the study of lipid behavior and molecular interactions. The cholesterol component of cholesteryl phenylacetate is in understanding the dynamics of lipid bilayers, as cholesterol is a fundamental component of cellular membranes. Its presence influences membrane fluidity, permeability, and the formation of lipid rafts, thereby providing insights into membrane-associated processes at the molecular level. The phenylacetate moiety, being aromatic, adds an additional dimension to the compound′s interactions, especially in terms of its hydrophobicity and potential for π-π interactions. This makes cholesteryl phenylacetate an important tool in probing the interactions of lipids with aromatic compounds, a scenario frequently encountered in the study of lipid-protein interactions and the behavior of lipid membranes in the presence of various small molecules.
Cholesteryl phenylacetate (CAS 33998-26-4) References
- Low Molecular Mass Gelators of Organic Liquids and the Properties of Their Gels. | Terech, P. and Weiss, RG. 1997. Chem Rev. 97: 3133-3160. PMID: 11851487
- Synthetic LXR agonists increase LDL in CETP species. | Groot, PH., et al. 2005. J Lipid Res. 46: 2182-91. PMID: 16024916
- Macrophage cholesteryl ester mobilization and atherosclerosis. | Ghosh, S., et al. 2010. Vascul Pharmacol. 52: 1-10. PMID: 19878739
- Replacement of endogenous cholesteryl esters of low density lipoprotein with exogenous cholesteryl linoleate. Reconstitution of a biologically active lipoprotein particle. | Krieger, M., et al. 1978. J Biol Chem. 253: 4093-101. PMID: 207690
- Numerical optimization of hydrothermal liquefaction process for the production of bio-oil and bio-char from Citrus maxima peel waste and product characterization. | Hussain, A., et al. 2023. Environ Sci Pollut Res Int. 30: 102462-102473. PMID: 37667127
- Oxidation of Cholesterol II. Influence of The Nature of The 3β‐Ester Group on The Yield of 17‐Ketosteroid | Maas S P J, van Dam M J D, De Heus J G, et al. 1963. Bulletin des Sociétés Chimiques Belges. 72: 239-254.
- The Odd-Even Effect in Steryl ω-Phenylalkanoates | Elser W, Pohlmann J L W, Boyd P R. 1971. Molecular Crystals and Liquid Crystals. 15: 175-182.
- Mesomorphism of homologous series. II. Odd-even effect | Ennulat R D, Brown A J. 1971. Molecular Crystals and Liquid Crystals. 12: 367-378.
- Methods development for high-speed preparative liquid chromatography | Larmann J P, Williams R C, Baker D R. 1975. Chromatographia. 8: 92-96.
- Gelation of silicone fluids using cholesteryl esters as gelators | Bujanowski V J, Katsoulis D E, Ziemelis M J. 1994. Journal of Materials Chemistry. 4: 1181-1187.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cholesteryl phenylacetate, 25 g | sc-214707 | 25 g | $1050.00 |
