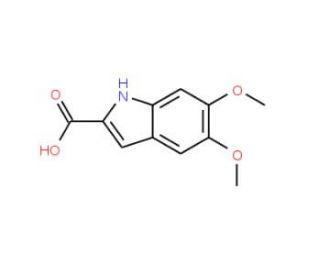
Molecular structure of 5,6-Dimethoxyindole-2-carboxylic acid, CAS Number: 88210-96-2
5,6-Dimethoxyindole-2-carboxylic acid (CAS 88210-96-2)
CAS Number:
88210-96-2
Molecular Weight:
221.21
Molecular Formula:
C11H11NO4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5,6-Dimethoxyindole-2-carboxylic acid is an organic compound firmly situated within the indole family. This natural substance is sourced from specific tree barks and holds a pivotal role in synthesizing various drugs and pharmaceuticals. Its utility isn′t confined to these realms alone it spans diverse applications within scientific research. From probing its biochemical and physiological impacts to playing a part in laboratory experiments, this compound′s significance is evident. Furthermore, its role isn′t limited to these arenas, as it has also contributed to the study of dopamine effects, further highlighting its versatility in unraveling intricate physiological pathways.
5,6-Dimethoxyindole-2-carboxylic acid (CAS 88210-96-2) References
- The Tyrosinase-tyrosine Reaction: Production from Tyrosine of 5: 6-Dihydroxyindole and 5: 6-Dihydroxyindole-2-carboxylic Acid-the Precursors of Melanin. | Raper, HS. 1927. Biochem J. 21: 89-96. PMID: 16743827
- The tyrosinase-tyrosine reaction: The action of tyrosinase on certain substances related to tyrosine. | Dulière, WL. and Raper, HS. 1930. Biochem J. 24: 239-49. PMID: 16744361
- Electrochromic conjugated polyheterocycles and derivatives--highlights from the last decade towards realization of long lived aspirations. | Gunbas, G. and Toppare, L. 2012. Chem Commun (Camb). 48: 1083-101. PMID: 21986913
- Alkene metathesis approach to β-unsubstituted anti-allylic alcohols and their use in ene-yne metathesis. | Clark, JR., et al. 2012. J Org Chem. 77: 1599-604. PMID: 22196022
- Biomaterials-based electronics: polymers and interfaces for biology and medicine. | Muskovich, M. and Bettinger, CJ. 2012. Adv Healthc Mater. 1: 248-66. PMID: 23184740
- Biologically derived melanin electrodes in aqueous sodium-ion energy storage devices. | Kim, YJ., et al. 2013. Proc Natl Acad Sci U S A. 110: 20912-7. PMID: 24324163
- Preparation of Thin Melanin-Type Films by Surface-Controlled Oxidation. | Salomäki, M., et al. 2016. Langmuir. 32: 4103-12. PMID: 27049932
- Triple Layer Tungsten Trioxide, Graphene, and Polyaniline Composite Films for Combined Energy Storage and Electrochromic Applications. | Lyu, H. 2019. Polymers (Basel). 12: PMID: 31905848
- Biological activity and synthesis of 5,6-dihydroxyindole-2-carboxylic acid - biosynthetic precursor of melanins (microreview). | Barabanov, MА., et al. 2021. Chem Heterocycl Compd (N Y). 57: 417-419. PMID: 34007085
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5,6-Dimethoxyindole-2-carboxylic acid, 1 g | sc-268149 | 1 g | $152.00 |
