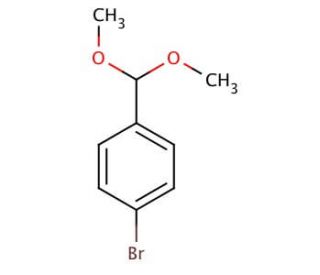
4-Bromobenzaldehyde dimethyl acetal (CAS 24856-58-4)
QUICK LINKS
4-Bromobenzaldehyde dimethyl acetal, known as 4-BBDA, is a versatile organic compound extensively utilized in diverse scientific research fields. This compound consists of a benzene-derived aldehyde with a bromine atom substitution at the 4-position and a dimethyl acetal group attached. It serves multiple roles, functioning as a reagent, catalyst, ligand, and building block for synthesizing various organic compounds. Displaying a faint odor, 4-Bromobenzaldehyde dimethyl acetal is a colorless liquid that exhibits excellent solubility in numerous organic solvents. Scientific research has embraced the myriad applications of 4-Bromobenzaldehyde dimethyl acetal. It finds extensive use in organic synthesis, catalysis, and materials science. As a reagent, it proves invaluable in the creation of diverse compounds, including pharmaceuticals, pesticides, and dyes. Additionally, its catalytic prowess enhances numerous reactions, exemplified by its involvement in the Suzuki-Miyaura coupling reaction. Furthermore, the compound serves as an essential building block in materials science for synthesizing polymers and various materials. The mechanism of action of 4-Bromobenzaldehyde dimethyl acetal varies according to its specific application. In organic synthesis, it serves as a nucleophile, initiating covalent bond formation by reacting with electrophiles. Within catalysis, the compound assumes a ligand role, binding to metal centers and stabilizing the transition state of reactions. In materials science, 4-Bromobenzaldehyde dimethyl acetal functions as a monomer, facilitating the linkage with other molecules to form polymers or diverse materials.
4-Bromobenzaldehyde dimethyl acetal (CAS 24856-58-4) References
- Synthesis of alpha- and beta-C-Aryl Delta(2)-Glycopyranosides from p-tert-Butylphenyl Delta(2)-Glycopyranosides via Grignard Reagents. | Moineau, C., et al. 1998. J Org Chem. 63: 582-591. PMID: 11672048
- p-Siletanylbenzylidene acetal: oxidizable protecting group for diols. | House, SE., et al. 2006. J Org Chem. 71: 420-2. PMID: 16388675
- The dihydrofuran template approach to furofuran synthesis. | Aldous, DJ., et al. 2006. Org Biomol Chem. 4: 2912-27. PMID: 16855740
- Large-scale applications of transition metal-catalyzed couplings for the synthesis of pharmaceuticals. | Magano, J. and Dunetz, JR. 2011. Chem Rev. 111: 2177-250. PMID: 21391570
- syn-Selective Kobayashi aldol reaction using acetals. | Tsukada, H., et al. 2013. Org Lett. 15: 678-81. PMID: 23331025
- Widely applicable metallacarborane reagents for π-conjugated systems. | Farràs, P., et al. 2014. Inorg Chem. 53: 5803-9. PMID: 24819777
- Visible light-promoted radical cyclization of silicon-tethered alkyl iodide and phenyl alkyne. An efficient approach to synthesize benzosilolines. | Lin, X., et al. 2016. Chem Commun (Camb). 52: 6189-92. PMID: 27078741
- Syn Selective Vinylogous Mukaiyama Aldol Reaction Using Z,E-Vinylketene N,O-Acetal with Acetals. | Sagawa, N., et al. 2017. Org Lett. 19: 250-253. PMID: 28001414
- Spatial acid-base-Pd triple-sites of a hierarchical core-shell structure for three-step tandem reaction. | Wu, J., et al. 2020. Chem Commun (Camb). 56: 6297-6300. PMID: 32386287
- Chemically Robust and Bifunctional Co(II)-Framework for Trace Detection of Assorted Organo-toxins and Highly Cooperative Deacetalization-Knoevenagel Condensation with Pore-Fitting-Induced Size-Selectivity. | Seal, N., et al. 2021. ACS Appl Mater Interfaces. 13: 28378-28389. PMID: 34100579
- Systematic Study of Regioselective Reductive Ring-Opening Reactions of 4,6-O-Halobenzylidene Acetals of Glucopyranosides. | Mezö, E., et al. 2021. J Org Chem. 86: 12973-12987. PMID: 34478619
- New aza-dipeptide analogues as potent and orally absorbed HIV-1 protease inhibitors: candidates for clinical development. | Bold, G., et al. 1998. J Med Chem. 41: 3387-401. PMID: 9719591
- Stereoelectronic effects in the DIBAL reduction of aryl-1,2-ethanediol benzylidene acetals | DR Gauthier Jr, RH Szumigala Jr, JD Armstrong III. 2001. Tetrahedron Letters. 42: 7011-7014.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Bromobenzaldehyde dimethyl acetal, 5 g | sc-226543 | 5 g | $40.00 |
