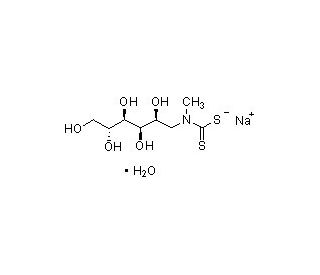
MGD sodium salt monohydrate 的分子结构, CAS编号: 91840-27-6
MGD sodium salt monohydrate (CAS 91840-27-6)
查看产品引用文献(5)
备用名:
N-(Dithiocarbamoyl)-N-methyl-D-glucamine•Na•H20; N-Methyl-D-glucamine dithiocarbamate; N-(D-Glucityl)-N-methyldithiocarbamate sodium salt
应用:
MGD sodium salt monohydrate是形成MGD2-Fe2+复合物的一种有用成分
CAS号码:
91840-27-6
纯度:
≥98%
分子量:
311.34
分子式:
C8H16NO5S2•Na•H2O
仅供科研使用。不可用于诊断或治疗。
* 参考分析证明 大量特定数据 (包括水 含量).
快捷链接
订购信息
产品相关文献索引
描述
技术信息
安全信息
安全技术说明书和分析证明书
MGD 钠盐单水合物是形成 MGD2-Fe2+ 复合物的有用成分,是一种极好的一氧化氮(NO)自旋捕获试剂。MGD2-Fe2+ 复合物不稳定,因此制成后应立即使用。为了形成与 FeSO4 的复合物,应使用过量的 MGD 来与 Fe2+ 形成更稳定的复合物溶液。应避免酸性条件,因为二硫代氨基甲酸盐容易分解形成有毒的二硫化碳。
MGD sodium salt monohydrate (CAS 91840-27-6) 参考文献
- 晚期肾病患者红细胞中的一氧化氮合成途径和 cGMP 水平发生了改变。 | Di Pietro, N., et al. 2016. Mol Cell Biochem. 417: 155-67. PMID: 27206740
- 用于癌症治疗的光控细菌代谢物。 | Zheng, DW., et al. 2018. Nat Commun. 9: 1680. PMID: 29700283
订购信息
| 产品名称 | 产品编号 | 规格 | 价格 | 数量 | 收藏夹 | |
MGD sodium salt monohydrate, 50 mg | sc-221941 | 50 mg | $88.00 | |||
MGD sodium salt monohydrate, 250 mg | sc-221941A | 250 mg | $251.00 |
