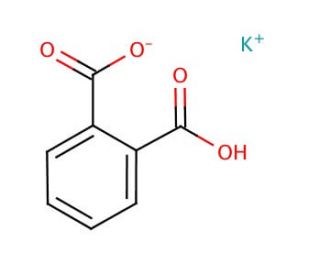
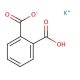
Potassium phthalate monobasic (CAS 877-24-7)
QUICK LINKS
Potassium phthalate monobasic is primarily utilized in analytical chemistry as a standard for calibrating pH meters and as a buffer solution in various chemical analyses. This compound effectively stabilizes pH levels, ensuring accuracy and consistency in experiments that are sensitive to pH fluctuations. Its buffering capacity is particularly valuable in colorimetric assays where precise pH control is essential to obtain reliable results. In addition to its role in pH management, potassium phthalate monobasic is also used in research to study the kinetic properties of enzymatic reactions that are pH-dependent. By providing a stable pH environment, this chemical helps in dissecting the influence of hydrogen ion concentration on enzyme activity and stability, thus contributing to a deeper understanding of enzyme mechanisms. Moreover, potassium phthalate monobasic finds application in the synthesis of phthalate polymers where it acts as a monomer. This aspect is explored in polymer science to develop materials with specific properties, such as flexibility, durability, and resistance to chemical degradation. The broad applicability of potassium phthalate monobasic in scientific research underscores its importance in enhancing methodological precision and expanding knowledge across disciplines, particularly in chemistry and materials science.
Potassium phthalate monobasic (CAS 877-24-7) References
- Characterization of phagolysosomal simulant fluid for study of beryllium aerosol particle dissolution. | Stefaniak, AB., et al. 2005. Toxicol In Vitro. 19: 123-34. PMID: 15582363
- Simultaneous speciation analysis of Sb(III), Sb(V) and (CH3)3SbCl2 by high performance liquid chromatography-hydride generation-atomic fluorescence spectrometry detection (HPLC-HG-AFS): application to antimony speciation in sea water. | De Gregori, I., et al. 2005. J Chromatogr A. 1091: 94-101. PMID: 16395797
- Quantification of Silicone Oil and Its Degradation Products in Aqueous Pharmaceutical Formulations by 1H-NMR Spectroscopy. | Malmstrøm, J. 2019. J Pharm Sci. 108: 1512-1520. PMID: 30471289
- A facile fabrication of dissolving microneedles containing 5-aminolevulinic acid. | Champeau, M., et al. 2020. Int J Pharm. 586: 119554. PMID: 32652182
- Self-Healing Hydrogel Scaffolds through PET-RAFT Polymerization in Cellular Environment. | Rigby, ADM., et al. 2023. Biomacromolecules. 24: 3370-3379. PMID: 37382088
- Protection of the right ventricular myocardium during acute right heart failure from pulmonary hypertension. | Albes, JM., et al. 1994. Eur Surg Res. 26: 62-8. PMID: 8137848
- Use of Tin (IV) Porhyrins as Ionophores for the Construction of Phthalate‐Selective Electrodes: Influence of the Structure and Membrane Composition on their Response Properties[J]. Electroanalysis: | Santos E M G, Araújo A N, Couto C M C M,. 2005. An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis. 17(21): 1945-1951.
- Wardak C. Ionic liquids as new lipophilic additives to the membrane of lead ion-selective electrodes with solid contact[J]. | . 2009. International Journal of Environmental and Analytical Chemistry,. 89(8-12): 735-748.
- Melamine-derived carbon electrode for efficient H2O2 electro-generation[J]. | Zhu Y, Qiu S, Ma F. 2018. Electrochimica Acta,. 261: 375-383.
- Assessment of the purity of emodin by quantitative nuclear magnetic resonance spectroscopy and mass balance[J]. | Park S, Choi Y J, Do G H,. 2019. Natural Product Sciences. 25(3): 222-227.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Potassium phthalate monobasic, 100 g | sc-203363 | 100 g | $41.00 | |||
Potassium phthalate monobasic, 500 g | sc-203363A | 500 g | $99.00 | |||
Potassium phthalate monobasic, 1 kg | sc-203363B | 1 kg | $179.00 | |||
Potassium phthalate monobasic, 2.5 kg | sc-203363C | 2.5 kg | $316.00 | |||
Potassium phthalate monobasic, 5 kg | sc-203363D | 5 kg | $571.00 |
