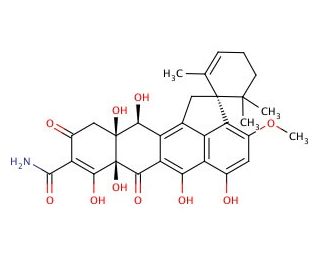
Viridicatumtoxin (CAS 39277-41-3)
QUICK LINKS
Viridicatumtoxin is an intriguing tetracycline-derived natural product, predominantly isolated from the fungus Penicillium aethiopicum. This compound has drawn significant attention in the research community due to its unique bicyclic structure characterized by a highly oxygenated aromatic system, which underpins its mode of action. The primary mechanism through which viridicatumtoxin operates involves the inhibition of bacterial protein synthesis. It achieves this by binding to the 30S ribosomal subunit, thereby blocking the attachment of aminoacyl-tRNA to the ribosomal acceptor (A) site, a critical step in protein translation. This action results in the cessation of bacterial growth, making it a valuable tool for studying bacterial protein synthesis and ribosome function. Research on viridicatumtoxin has expanded to explore its potential role as a lead compound in the development of new bioactive molecules. Its ability to inhibit protein synthesis in bacteria, coupled with its unique chemical structure, makes it an excellent candidate for chemical modification and optimization studies aimed at enhancing its bioactivity and specificity. Investigations into viridicatumtoxin also include its interactions with other cellular targets and pathways, particularly looking at its effects on bacterial physiology and potential applications in controlling bacterial populations in various environmental and industrial settings.
Viridicatumtoxin (CAS 39277-41-3) References
- Viridicatumtoxin B, a new anti-MRSA agent from Penicillium sp. FR11. | Zheng, CJ., et al. 2008. J Antibiot (Tokyo). 61: 633-7. PMID: 19168978
- Studies of the teratogenicity of xanthomegnin and viridicatumtoxin in ICR mice. | Bolin, DC., et al. 1991. Toxicol Lett. 55: 273-7. PMID: 2003269
- Identification of the viridicatumtoxin and griseofulvin gene clusters from Penicillium aethiopicum. | Chooi, YH., et al. 2010. Chem Biol. 17: 483-94. PMID: 20534346
- [Mode of action of microbial anti-MRSA agents]. | Tomoda, H. 2012. Yakugaku Zasshi. 132: 37-44. PMID: 22214578
- Spirohexalines, new inhibitors of bacterial undecaprenyl pyrophosphate synthase, produced by Penicillium brasilianum FKI-3368. | Inokoshi, J., et al. 2013. J Antibiot (Tokyo). 66: 37-41. PMID: 23168407
- A genomics based discovery of secondary metabolite biosynthetic gene clusters in Aspergillus ustus. | Pi, B., et al. 2015. PLoS One. 10: e0116089. PMID: 25706180
- Inhibition of bacterial undecaprenyl pyrophosphate synthase by small fungal molecules. | Inokoshi, J., et al. 2016. J Antibiot (Tokyo). 69: 798-805. PMID: 27049441
- Investigations into the Antibacterial Mechanism of Action of Viridicatumtoxins. | Li, W., et al. 2020. ACS Infect Dis. 6: 1759-1769. PMID: 32437130
- Viridicatumtoxin, a new mycotoxin from Penicillium viridicatum Westling. | Hutchison, RD., et al. 1973. Toxicol Appl Pharmacol. 24: 507-9. PMID: 4122267
- Viridicatumtoxin mycotoxicosis in mice and rats. | Bendele, AM., et al. 1984. Toxicol Lett. 22: 287-91. PMID: 6485002
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Viridicatumtoxin, 1 mg | sc-391505 | 1 mg | $280.00 |
