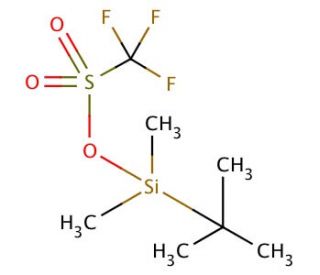
tert-Butyldimethylsilyl trifluoromethanesulfonate (CAS 69739-34-0)
QUICK LINKS
Tert-butyldimethylsilyl trifluoromethanesulfonate (TBDMS-Tf) is an organosilicon compound widely employed as an electrophilic reagent for introducing t-butyldimethylsilyl groups. It is a colorless liquid, soluble in various organic solvents such as ether, acetone, and chloroform. Additionally, Tert-butyldimethylsilyl trifluoromethanesulfonate exhibits solubility in water, possessing a higher boiling point compared to most organic solvents. With its diverse applications in organic synthesis, Tert-butyldimethylsilyl trifluoromethanesulfonate finds extensive use in the creation of various compounds, including amines, alcohols, and other organosilicon derivatives. Primarily, it serves as a reagent for the selective protection of alcohols, amines, and functional groups in organic synthesis. Moreover, Tert-butyldimethylsilyl trifluoromethanesulfonate plays a role in peptide synthesis and the development of biologically active molecules. Furthermore, it facilitates the synthesis of an array of organosilicon compounds such as silanes, siloxanes, and silanes. Tert-butyldimethylsilyl trifluoromethanesulfonate acts as a nucleophile, targeting electrophilic centers within organic molecules. The reaction occurs rapidly, and the resulting product is isolated through distillation. It is important to note that Tert-butyldimethylsilyl trifluoromethanesulfonate does not exhibit significant biochemical or physiological effects. It is considered non-toxic, causing no adverse reactions in humans or animals.
tert-Butyldimethylsilyl trifluoromethanesulfonate (CAS 69739-34-0) References
- 2'-C-Branched Ribonucleosides: Synthesis of the Phosphoramidite Derivatives of 2'-C-beta-Methylcytidine and Their Incorporation into Oligonucleotides. | Tang, XQ., et al. 1999. J Org Chem. 64: 747-754. PMID: 11674142
- Improved synthesis of 5-hydroxylysine (Hyl) derivatives. | Cudic, M., et al. 2005. J Pept Res. 65: 272-83. PMID: 15705169
- A convergent enantioselective route to structurally diverse 6-deoxytetracycline antibiotics. | Charest, MG., et al. 2005. Science. 308: 395-8. PMID: 15831754
- Syntheses of biologically active natural products and leading compounds for new pharmaceuticals employing effective construction of a polycyclic skeleton. | Ihara, M. 2006. Chem Pharm Bull (Tokyo). 54: 765-74. PMID: 16755041
- Radical alkylation of bis(silyloxy)enamine derivatives of organic nitro compounds. | Lee, JY., et al. 2006. Angew Chem Int Ed Engl. 45: 6182-6. PMID: 16906611
- Synthesis and antibacterial activity of 3-keto-6-O-carbamoyl-11,12-cyclic thiocarbamate erythromycin A derivatives. | Zhu, B., et al. 2007. Bioorg Med Chem Lett. 17: 3900-4. PMID: 17502142
- Synthesis and solid-phase application of suitably protected gamma-hydroxyvaline building blocks. | Cudic, M., et al. 2007. J Org Chem. 72: 5581-6. PMID: 17583956
- Synthesis of photosynthesis-inhibiting nostoclide analogues. | Teixeira, RR., et al. 2008. J Agric Food Chem. 56: 2321-9. PMID: 18338868
- The total synthesis of D-chalcose and its C-3 epimer. | Sun, J., et al. 2013. Beilstein J Org Chem. 9: 2620-4. PMID: 24367425
- Rearrangement of a Transient Gold Vinylidene into Gold Carbenes. | Debrouwer, W. and Fürstner, A. 2017. Chemistry. 23: 4271-4275. PMID: 28230280
- Profiling Vaccinium macrocarpon components and metabolites in human urine and the urine ex-vivo effect on Candida albicans adhesion and biofilm-formation. | Baron, G., et al. 2020. Biochem Pharmacol. 173: 113726. PMID: 31778647
- Syntheses and reactions of silyl carbamates. 1. Chemoselective transformation of amino protecting groups via tert-butyldimethylsilyl carbamates | Masahiro Sakaitani and Yasufumi Ohfune. 1990,. J. Org. Chem. 55, 3,: 870–876.
- Reaction of thiophenol with glucal epoxides: X-ray structure of 3,4,6-tri-O-tert-butyldimethylsilyl-1-S-phenyl-1-thio-α-D-glucopyranoside | . 1997,. Chem. Commun.,. 1855-1856.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
tert-Butyldimethylsilyl trifluoromethanesulfonate, 1 g | sc-251141 | 1 g | $28.00 | |||
tert-Butyldimethylsilyl trifluoromethanesulfonate, 5 g | sc-251141A | 5 g | $62.00 | |||
tert-Butyldimethylsilyl trifluoromethanesulfonate, 100 g | sc-251141B | 100 g | $118.00 | |||
tert-Butyldimethylsilyl trifluoromethanesulfonate, 500 g | sc-251141C | 500 g | $407.00 |
