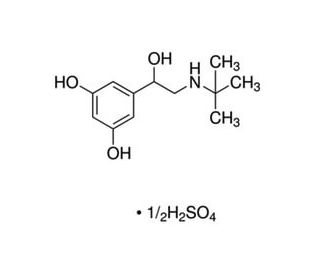
Terbutaline Hemisulfate 의 분자 구조, CAS 번호: 23031-32-5
Terbutaline Hemisulfate (CAS 23031-32-5)
인용논문 보기 (2)
대체 이름:
Bricanyl
적용:
Terbutaline Hemisulfate 은 β2- 아드레날린 수용체의 작용제입니다.
CAS 등록번호:
23031-32-5
순도:
≥98%
분자량:
274.32
분자식:
C12H19NO3•1/2(H2SO4)
연구용으로만 사용가능합니다. 진단이나 치료용으로 사용불가합니다.
* 참조분석증명서대량의 측정 데이터(함수량포함).
빠른 링크
주문정보
논문정보
설명
기술정보
안전정보
SDS 및 분석 증명서
테르부탈린은 기관지 확장제로 작용하는 β2- 아드레날린 수용체 작용제입니다. 기도의 평활근 세포에 있는 β2- 아드레날린 수용체에 결합하여 아데닐레이트 시클라제를 활성화하고 이후 세포 내 순환 AMP 수준을 증가시키는 작용을 합니다. 그 결과 기관지 평활근이 이완되어 기관지 확장 및 폐의 공기 흐름이 개선됩니다. 테르부탈린은 기관지 수축을 유발할 수 있는 히스타민 및 류코트리엔과 같은 비만 세포의 염증 매개 물질 방출을 억제할 수 있습니다. 분자 수준에서 테르부탈린과 β2 아드레날린 수용체와의 상호작용은 일련의 세포 내 사건으로 이어져 궁극적으로 기관지 평활근의 이완을 초래하므로 실험에서 기도 평활근 긴장도 조절을 연구하는 데 사용할 수 있습니다.
Terbutaline Hemisulfate (CAS 23031-32-5) 참고자료
- 고성능 액체 크로마토그래피로 테르부탈린 혈장 농도를 측정하는 간단한 방법. | Herring, VL. and Johnson, JA. 2000. J Chromatogr B Biomed Sci Appl. 741: 307-12. PMID: 10872601
- 저산소증과 베타 2- 작용제는 원시 폐포 상피 세포에서 상피 나트륨 채널의 세포 표면 발현을 조절합니다. | Planès, C., et al. 2002. J Biol Chem. 277: 47318-24. PMID: 12372821
- 테르부탈린과 클로르피리포스에 대한 발달 노출: 조기 진통의 약물 요법과 환경 신경 독성 물질이 신생아 쥐의 뇌 영역에서 세로토닌 시스템에 수렴합니다. | Aldridge, JE., et al. 2005. Toxicol Appl Pharmacol. 203: 132-44. PMID: 15710174
- 베타2- 아드레날린 수용체: 베타2- 작용제의 작용 메커니즘. | Johnson, M. 2001. Paediatr Respir Rev. 2: 57-62. PMID: 16263481
- 테르부탈린은 인간 영양막 세포에서 코르티코트로핀 방출 호르몬(CRH) 발현을 억제합니다. | Kontopoulos, EV., et al. 2006. J Matern Fetal Neonatal Med. 19: 735-9. PMID: 17127497
- 쥐의 신생아 테르부탈린 치료 후 신경 염증 및 행동 이상: 자폐증에 대한 시사점. | Zerrate, MC., et al. 2007. J Pharmacol Exp Ther. 322: 16-22. PMID: 17400887
- 베타(2)-길항제는 비정형적인 cAMP 의존 경로를 통해 기니피그의 투쟁성 반응을 차단합니다. | Freund-Michel, VC., et al. 2010. Eur Respir J. 35: 647-54. PMID: 19679606
- 항우울제는 말초 β2- 아드레날린 수용체 매개 항-TNFα 기전을 통해 신경병증성 통증을 억제합니다. | Bohren, Y., et al. 2013. Neurobiol Dis. 60: 39-50. PMID: 23978467
- 제브라피쉬의 운동 활성 분석에 기반한 약물 유발 발작 책임 스크리닝을 위한 새로운 실험 프로토콜을 수립합니다. | Koseki, N., et al. 2014. J Toxicol Sci. 39: 579-600. PMID: 25056783
- 간질 상태는 쥐의 상아교근 분자층에서 성숙한 과립 세포를 증가시킵니다. | Liang, Z., et al. 2013. Neural Regen Res. 8: 609-15. PMID: 25206705
- 쥐에서 필로카르핀으로 유도된 간질 상태 후 태어난 성숙한 과립 세포의 수지상 형태, 시냅스 전달 및 활성. | Gao, F., et al. 2015. Front Cell Neurosci. 9: 384. PMID: 26500490
- 조건부 NeuroD1 녹아웃 마우스는 비정상적인 해마 신경 생성이 감소했지만 간질 발작에는 변화가 없었습니다. | Brulet, R., et al. 2017. Exp Neurol. 293: 190-198. PMID: 28427858
- 쥐에서 천식 약물에 의한 급성 심근 병변의 향상. | Whitehurst, VE., et al. 1994. Toxicol Pathol. 22: 72-6. PMID: 7915431
- 테르부탈린 흡입은 펜타닐로 인한 기침을 억제합니다. | Lui, PW., et al. 1996. Can J Anaesth. 43: 1216-9. PMID: 8955969
- 밀리그램 양의 테르부탈린의 키랄 분리에 고전적인 겔 전기영동을 적용합니다. | Stalcup, AM., et al. 1998. Anal Chem. 70: 144-8. PMID: 9435473
의 억제제:
beta1-AR.의 활성화자:
β2-AR, 1190002N15Rik, 1700034O15Rik, Ag2, APBA2BP, ARHGAP20, ARHGAP4, ARHGAP9, ARRDC2, ATP9BL, ATP9BL2, BAALC, Basal ciliated, Basonuclin 2, beta2-AR, C11orf70, C14orf143, C19orf35, CCDC105, CCDC153, CCDC60_Ccdc60, CCDC82, CCDC84, CCDC85C, CCDC9, CCDC91, CCDC94, CCDC97, CELSR3, CHSY2, CLDND2, CREBL2, D54, EG628705, ENDOD1, eoxtaxin-3, ETL, FAM118B, FAM228B_A830093I24Rik, GDE6, GHRH-R, Gm129, GNPTAB, GPATCH1, GPR-137, GPR-152, H2-Q1, HARBI1, HuC, KBTBD3, KCTD21, KDELC2, KLHL26, LMO3, LOC388564, MBD3L1, MBD3L5, Med29, METT5D1, MIPU1, MORN3, MPND, MRP-L34, MS4A13, MS4A3, MS4A6A, MYLPF, Nim1, NPS, NRK, OCEL1, Odf3, Olr1020, Olr104, Olr1059, Olr1061, Olr434, OR10A5, OR4A16, OR4C13, OR4C3, OR4D9, OR4P4, OR4X1, OR56A4, OR5B12, OR6K2, OR8B12, OR8G5, OR9Q2, OVCH1, OVCH2, PAAF1, PAC4, PC1, PDE7B, PGT, PHACTR2, PHKG2, PIH1D2, PLEKHH2, PRR9, PSG7, R9AP, Ramp4, REP15, RERGL, RFPL4B, RSHL1, Sarcalumenin, SCGB1D4, SDF-2L1, SEC14L1, sFlt-1, Skint9, SNX10, SPA-L3, SPRED2, SULT2A3, SULT2A6, TBCEL, TCP-10b, Tdpoz2, Tdpoz3, Tdpoz5, TDRD10, TDRD7, Tect1, Tenrl, TEX10, TEX16, TEX19.2, TMEM86B, TMEM9B, TPSB3, TRPT1, TTC9C, UBQLNL, V1RA4, V3R 2, VMAC, Vmn2r6, Vmn2r60, Vmn2r63, Vmn2r66, Vmn2r68, XLαs, YPEL4, ZFPL1.주문정보
| 제품명 | 카탈로그 번호 | 단위 | 가격 | 수량 | 관심품목 | |
Terbutaline Hemisulfate, 1 g | sc-204911 | 1 g | $92.00 | |||
Terbutaline Hemisulfate, 5 g | sc-204911A | 5 g | $378.00 |
