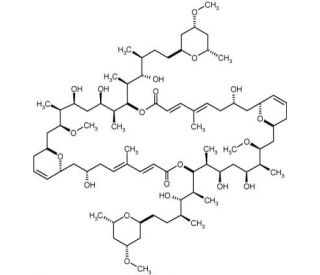
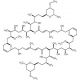
Swinholide A, Theonella swinhoei (CAS 95927-67-6)
QUICK LINKS
Swinholide A, a natural product isolated from the marine sponge Theonella swinhoei, is a potent cytotoxic macrolide known for its unique mechanism of disrupting the actin cytoskeleton. Swinholide A exerts its effects by binding to actin filaments, promoting their severing and leading to the depolymerization of the actin network. This disruption of the cytoskeleton affects various cellular processes, including cell shape, motility, and division, making it a valuable tool in cell biology research. In research, Swinholide A is used to study the dynamics and regulation of the actin cytoskeleton. Its ability to sever actin filaments has been instrumental in elucidating the roles of actin in maintaining cellular architecture and in processes such as intracellular transport and signal transduction. Researchers employ Swinholide A to investigate the molecular mechanisms underlying actin filament turnover and to identify the pathways and proteins involved in cytoskeletal regulation. Furthermore, Swinholide A serves as a model compound for studying the interactions between natural products and the cytoskeleton, providing insights into how marine-derived macrolides can influence cellular functions. Its use in high-content screening assays helps identify potential modulators of actin dynamics, contributing to the broader understanding of cytoskeletal biology. The research applications of Swinholide A extend to exploring its impact on cellular processes.
Swinholide A, Theonella swinhoei (CAS 95927-67-6) References
- Structural basis of swinholide A binding to actin. | Klenchin, VA., et al. 2005. Chem Biol. 12: 287-91. PMID: 15797212
- Highlights of marine invertebrate-derived biosynthetic products: their biomedical potential and possible production by microbial associants. | Radjasa, OK., et al. 2011. Bioorg Med Chem. 19: 6658-74. PMID: 21835627
- Early-phase drug discovery of β-III-spectrin actin-binding modulators for treatment of spinocerebellar ataxia type 5. | Guhathakurta, P., et al. 2023. J Biol Chem. 299: 102956. PMID: 36731793
- PHACTR1 promotes the mobility of papillary thyroid carcinoma cells by inducing F-actin formation. | Zang, L., et al. 2023. Heliyon. 9: e20461. PMID: 37876444
- Actin Isoform Composition and Binding Factors Fine-Tune Regulatory Impact of Mical Enzymes. | Martin, JL., et al. 2023. Int J Mol Sci. 24: PMID: 38068973
- Development of actin dimerization inducers inspired by actin-depolymerizing macrolides. | Itakura, M., et al. 2024. Chem Commun (Camb). 60: 4910-4913. PMID: 38623638
- Swinholide A is a microfilament disrupting marine toxin that stabilizes actin dimers and severs actin filaments. | Bubb, MR., et al. 1995. J Biol Chem. 270: 3463-6. PMID: 7876075
- Marine natural products. XXXI. Structure-activity correlation of a potent cytotoxic dimeric macrolide swinholide A, from the Okinawan marine sponge Theonella swinhoei, and its isomers. | Kobayashi, M., et al. 1994. Chem Pharm Bull (Tokyo). 42: 19-26. PMID: 8124763
- Two classes of metabolites from Theonella swinhoei are localized in distinct populations of bacterial symbionts. | Bewley, CA., et al. 1996. Experientia. 52: 716-22. PMID: 8698116
- Autoregulation of actin synthesis responds to monomeric actin levels. | Lyubimova, A., et al. 1997. J Cell Biochem. 65: 469-78. PMID: 9178097
- Actin-depolymerizing effect of dimeric macrolides, bistheonellide A and swinholide A. | Saito, SY., et al. 1998. J Biochem. 123: 571-8. PMID: 9538245
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Swinholide A, Theonella swinhoei, 10 µg | sc-205914 | 10 µg | $135.00 |
