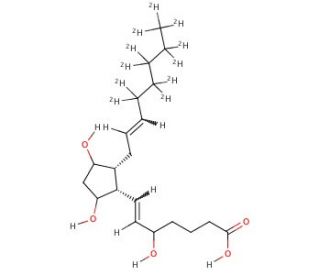
(±)5-iPF2α-VI-d11 (CAS 936565-17-2)
QUICK LINKS
5-iPF2α-VI-d11 contains 11 deuterium atoms and is intended for use as an internal standard for quantification of 5-iPF2α-VI (sc-205138) by GC- or LC-mass spectrometry. Isoprostanes are prostaglandin-like products of free-radical induced lipid peroxidation. iPF2α-VI is one of dozens of possible stereo- and regioisomeric isoprostanes that can be formed from arachidonic acid. As of now, the most extensively studied of these is 8-isoprostane (8-epi-PGF2α, iPF2α-III) (sc-201261). However, 8-isoprostane is a minor isoprostane constituent when compared to some of the other isomers that form in natural conditions of oxidative stress, including iPF2α-VI of the type-VI isoprostanes. This class has been shown to be one of the major isoprostane products, in contrast to 8-isoprostane. In addition to being produced in greater abundance than 8-isoprostane, Type VI isoprostanes form internal lactones which facilitate their extraction and purification from biological samples.
(±)5-iPF2α-VI-d11 (CAS 936565-17-2) References
- Quantitative high performance liquid chromatography/tandem mass spectrometric analysis of the four classes of F(2)-isoprostanes in human urine. | Li, H., et al. 1999. Proc Natl Acad Sci U S A. 96: 13381-6. PMID: 10557329
- A series of prostaglandin F2-like compounds are produced in vivo in humans by a non-cyclooxygenase, free radical-catalyzed mechanism. | Morrow, JD., et al. 1990. Proc Natl Acad Sci U S A. 87: 9383-7. PMID: 2123555
- Chemical structure and sweet taste of isocoumarin and related compounds. VII. | Yamato, M., et al. 1977. Chem Pharm Bull (Tokyo). 25: 695-9. PMID: 880713
- IPF2alpha-I: an index of lipid peroxidation in humans. | Praticò, D., et al. 1998. Proc Natl Acad Sci U S A. 95: 3449-54. PMID: 9520386
- Identification of two major F2 isoprostanes, 8,12-iso- and 5-epi-8, 12-iso-isoprostane F2alpha-VI, in human urine. | Lawson, JA., et al. 1998. J Biol Chem. 273: 29295-301. PMID: 9792628
- Evidence for the formation of dinor isoprostanes E1 from alpha-linolenic acid in plants. | Parchmann, S. and Mueller, MJ. 1998. J Biol Chem. 273: 32650-5. PMID: 9830005
- Increased formation of distinct F2 isoprostanes in hypercholesterolemia. | Reilly, MP., et al. Circulation. 98: 2822-8. PMID: 9860782
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(±)5-iPF2α-VI-d11, 10 µg | sc-221031 | 10 µg | $600.00 | |||
(±)5-iPF2α-VI-d11, 25 µg | sc-221031A | 25 µg | $1405.00 |
