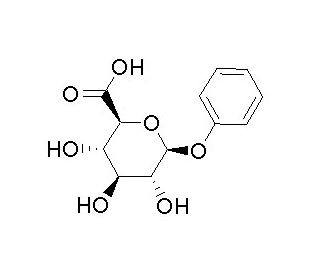
Phenyl-β-D-glucuronide 的分子结构, CAS编号: 17685-05-1
Phenyl-β-D-glucuronide (CAS 17685-05-1)
备用名:
Phenyl-β-D-glucopyranosiduronic acid
应用:
Phenyl-β-D-glucuronide 是一种 β-葡萄糖醛酸酶底物
CAS号码:
17685-05-1
纯度:
≥98%
分子量:
270.24
分子式:
C12H14O7
仅供科研使用。不可用于诊断或治疗。
* 参考分析证明 大量特定数据 (包括水 含量).
快捷链接
订购信息
描述
技术信息
安全信息
安全技术说明书和分析证明书
苯基-β-D-葡糖苷酸,简称苯基葡糖苷酸(PG),是一种代谢产物。在实验室环境中,苯基-β-D-葡糖苷酸可以通过在硫酸存在下使葡糖醛酸与苯酚反应合成,产生苯基-β-D-葡糖苷酸和水作为副产物。高效液相色谱法(HPLC)可用于测定合成的PG的纯度。苯基-β-D-葡糖苷酸的形成是通过葡糖苷酸化,这是一个由称为UDP-葡糖苷酸转移酶(UGT)的酶催化的过程。
Phenyl-β-D-glucuronide (CAS 17685-05-1) 参考文献
- 高效液相色谱法检测人体尿液中丙泊酚的羟酸盐和共轭代谢物。 | Favetta, P., et al. 2000. J Chromatogr B Biomed Sci Appl. 742: 25-35. PMID: 10892581
- 大肠杆菌的 gusBC 基因编码葡萄糖醛酸转运系统。 | Liang, WJ., et al. 2005. J Bacteriol. 187: 2377-85. PMID: 15774881
- 利用固相萃取-液相色谱-串联质谱法定量测定人体血浆中的异丙酚及其主要葡萄糖醛酸代谢物。 | Cohen, S., et al. 2007. J Chromatogr B Analyt Technol Biomed Life Sci. 854: 165-72. PMID: 17485254
- LC-MS兼容HPLC分离异种生物及其I期和II期代谢物:阴离子交换和反相色谱同时进行。 | Johnston, JJ., et al. 1991. J Chromatogr Sci. 29: 511-6. PMID: 1761635
- 大肠杆菌葡萄糖醛酸合成酶:一种用于合成 beta-葡萄糖醛酸的工程酶。 | Wilkinson, SM., et al. 2008. Org Lett. 10: 1585-8. PMID: 18345681
- 药物分析中的 HILIC 方法。 | Dejaegher, B. and Vander Heyden, Y. 2010. J Sep Sci. 33: 698-715. PMID: 20183826
- 大肠杆菌葡萄糖醛酸合成酶的实验和动力学研究:一种用于合成葡萄糖醛酸共轭物的工程酶。 | Wilkinson, SM., et al. 2011. J Org Chem. 76: 1992-2000. PMID: 21348473
- 比较分析尿液中丙泊酚及其代谢物的气相色谱/质谱法和液相色谱/质谱法。 | Lee, SY., et al. 2012. J Chromatogr B Analyt Technol Biomed Life Sci. 900: 1-10. PMID: 22672847
- 利用糖苷合成金纳米粒子:基于糖醛和糖醛酸结构的合成趋势。 | Jung, J., et al. 2014. Carbohydr Res. 386: 57-61. PMID: 24491842
- 简便灵敏的气相色谱/质谱法,用于定量检测尿液中的苯酚, 邻甲酚, 间甲酚和乙基苯酚,作为暴露于工业溶剂的生物标志物。 | Schettgen, T., et al. 2015. J Chromatogr B Analyt Technol Biomed Life Sci. 995-996: 93-100. PMID: 26046979
- 熊果苷-金纳米粒子复合物的合成及其增强的美白性能。 | Park, JJ., et al. 2019. Arch Pharm Res. 42: 977-989. PMID: 31144234
- 尿毒症环境对慢性肾脏病中促炎单核细胞表型增加的贡献。 | Borges Bonan, N., et al. 2019. Sci Rep. 9: 10236. PMID: 31308443
- MRP4 负责将霉酚酸 β-d 葡萄糖醛酸(MPAG)从肝细胞外排到血液中。 | Berthier, J., et al. 2021. Xenobiotica. 51: 105-114. PMID: 32820679
的基质:
β-glucuronidase, Chromogenic, 和 Enzyme.订购信息
| 产品名称 | 产品编号 | 规格 | 价格 | 数量 | 收藏夹 | |
Phenyl-β-D-glucuronide, 25 mg | sc-222163 | 25 mg | $50.00 | |||
Phenyl-β-D-glucuronide, 100 mg | sc-222163A | 100 mg | $144.00 |
