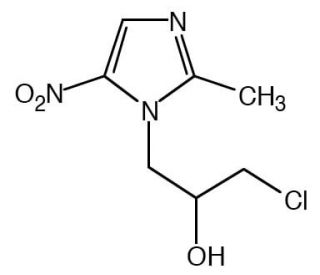
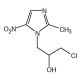
Ornidazole (CAS 16773-42-5)
QUICK LINKS
Ornidazole, identified by the CAS number 16773-42-5, is a synthetic derivative within the class of 5-nitroimidazoles, known for its activity against protozoa and anaerobic bacteria. The chemical acts by entering microbial cells and getting reduced by low redox potential electron-transport proteins, which are more abundant in anaerobic conditions. This reduction process leads to the formation of toxic intermediates and free radicals, which subsequently damage microbial DNA and other critical cellular structures, thus inhibiting further replication and transcription in the microorganisms. In research contexts, Ornidazole has been extensively utilized to study the mechanisms of selective toxicity towards anaerobes and protozoal cells, providing insights into the molecular basis of its action and the resistance patterns that can evolve. Researchers also use Ornidazole to explore the interaction between microbial metabolism and drug efficacy, especially focusing on the role of the nitroreductase enzymes that activate the nitroimidazole moiety. This research is crucial for understanding the dynamics of microbial communities under selective pressure from nitroimidazole compounds and helps in studying the biochemical pathways involved in microbial pathogenicity and survival in anaerobic environments.
Ornidazole (CAS 16773-42-5) References
- Nanomanganese cobaltate-decorated halloysite nanotubes for the complete degradation of ornidazole via peroxymonosulfate activation. | Zhang, Y., et al. 2023. J Colloid Interface Sci. 630: 855-866. PMID: 36283268
- Ornidazole suppresses CD133+ melanoma stem cells via inhibiting hedgehog signaling pathway and inducing multiple death pathways in a mouse model. | Evyapan, G., et al. 2022. Croat Med J. 63: 461-474. PMID: 36325671
- Green synthesis, characterization, and antimicrobial applications of silver nanoparticles as fluorescent nanoprobes for the spectrofluorimetric determination of ornidazole and miconazole. | Magdy, G., et al. 2022. Sci Rep. 12: 21395. PMID: 36496441
- Low-temperature molten-salt synthesis of Co3O4 nanoparticles grown on MXene can rapidly remove ornidazole via peroxymonosulfate activation. | Hao, C., et al. 2023. Environ Pollut. 334: 121811. PMID: 37209900
- Electrochemical detection of ornidazole in commercial milk and water samples using an electrode based on green synthesis of silver nanoparticles using cellulose separated from Phoenix dactylifera seed. | Ettadili, FE., et al. 2023. Int J Biol Macromol. 242: 124995. PMID: 37236559
- Physicochemical considerations in the formulation development of silicone elastomer vaginal rings releasing 5-nitroimidazole drugs for the treatment of bacterial vaginosis. | Zhao, X., et al. 2023. Int J Pharm. 644: 123296. PMID: 37553058
- Tong Jing Yi Hao Formula Alleviates Ornidazole-Induced Oligoasthenospermia in Rats by Suppressing ROS/MAPK/HIF-1 Pathway. | Tian, Z., et al. 2023. Arch Esp Urol. 76: 596-604. PMID: 37960959
- Kinetic constants and transformation products of ornidazole during ozonation. | Fan, S., et al. 2024. Chemosphere. 349: 140783. PMID: 38043618
- Long-term stable and efficient degradation of ornidazole with minimized by-product formation by a biological sulfidogenic process based on elemental sulfur. | Zeng, J., et al. 2024. Water Res. 249: 120940. PMID: 38071904
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ornidazole, 5 g | sc-205779 | 5 g | $51.00 | |||
Ornidazole, 50 g | sc-205779A | 50 g | $460.00 |
