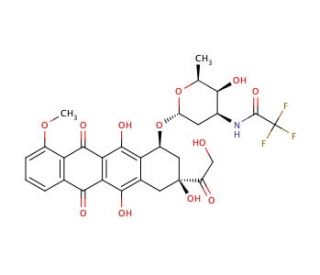
N-Trifluoroacetyldoxorubicin (CAS 26295-56-7)
QUICK LINKS
N-Trifluoroacetyldoxorubicin is a chemically modified analogue of doxorubicin, distinguished by the addition of a trifluoroacetyl group. This modification aims to explore and understand the fundamental chemical and biochemical properties of the molecule, particularly how such modifications affect its interaction with biological macromolecules. The primary mechanism of action of N-Trifluoroacetyldoxorubicin involves intercalating into DNA, a process where the molecule inserts itself between the base pairs of the DNA helix. This intercalation disrupts the normal function of the DNA by blocking the replication and transcription processes. The trifluoroacetyl group potentially enhances the lipophilicity of the molecule, which may influence its ability to cross cellular membranes and its overall stability within biological environments. In scientific research, N-Trifluoroacetyldoxorubicin has been utilized to study the effects of chemical modifications on the behavior of anthracycline antibiotics, focusing on their interaction with DNA and other nucleic acids. This research is crucial for elucidating the structural features that govern the activity and specificity of these molecules. Additionally, studies involving N-Trifluoroacetyldoxorubicin contribute to broader understanding of the dynamics of DNA interaction with small molecules, providing insights that are valuable for the field of molecular biology and chemistry.
N-Trifluoroacetyldoxorubicin (CAS 26295-56-7) References
- Bacillus calmette-guérin failures and beyond: contemporary management of non-muscle-invasive bladder cancer. | Grossman, HB., et al. 2008. Rev Urol. 10: 281-9. PMID: 19145271
- The mechanism of action of quinone antibiotics. | Lown, JW. 1983. Mol Cell Biochem. 55: 17-40. PMID: 6353197
- Radioactive species in rat urines and tissues after [14C] AD 32 administration. | Zini, G., et al. 1984. Cancer Chemother Pharmacol. 12: 154-6. PMID: 6705133
- Anthracycline antibiotic blockade of SV40 T antigen helicase action. | Bachur, NR., et al. 1998. Biochem Pharmacol. 55: 1025-34. PMID: 9605426
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Trifluoroacetyldoxorubicin, 5 mg | sc-391221 | 5 mg | $360.00 |
