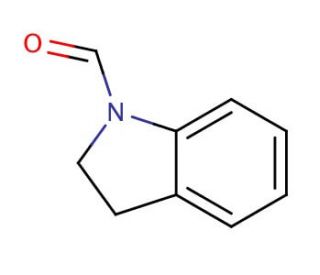
N-Formylindoline (CAS 2861-59-8)
LINKS RÁPIDOS
A N-Formilindolina é um composto orgânico que desempenha um papel importante na química sintética, particularmente na síntese de vários compostos heterocíclicos. Este produto químico é conhecido pela sua estrutura distintiva, em que o grupo formilo ligado ao azoto do anel de indolina o torna um intermediário valioso na síntese orgânica. O principal mecanismo de ação da N-Formilindolina envolve a sua participação em reacções de condensação, onde actua como agente formilante. O grupo formilo é altamente reativo e pode facilitar a formação de ligações carbono-nitrogénio ao reagir com aminas, levando à síntese de bases de schiff e outros compostos de imina. Esta reatividade é crucial para as modificações da estrutura da indolina, que é uma estrutura comum em muitos produtos naturais e moléculas sintéticas. Na investigação, a N-Formilindolina tem sido utilizada para estudar a síntese de moléculas orgânicas complexas, incluindo análogos de produtos naturais e produtos farmacêuticos. A sua utilidade na construção de diversas arquitecturas moleculares advém da sua capacidade de sofrer reacções de adição nucleofílica com vários nucleófilos, levando à formação de uma vasta gama de indolinas funcionalizadas. Além disso, a N-Formilindolina está envolvida no estudo de processos catalíticos em que o seu grupo formilo pode sofrer transformações selectivas sob diferentes condições catalíticas. Os investigadores exploraram o seu comportamento sob catálise de metais de transição, o que pode levar a uma funcionalização selectiva de C-H ou a reacções de acoplamento cruzado. Estes estudos ajudam a desenvolver novas metodologias para a construção eficiente de moléculas complexas.
N-Formylindoline (CAS 2861-59-8) Referencias
- Preparação de 7-Halo-indóis por Talação de N-formilindolina e sua tentativa de utilização para a síntese do segmento da mão direita da cloropeptina. | Yamada, Y., et al. 2006. Chem Pharm Bull (Tokyo). 54: 788-94. PMID: 16755045
- Avanços recentes na N-formilação catalítica de aminas com CO2 mediada por hidrosilano líquido. | Li, Z., et al. 2020. RSC Adv. 10: 33972-34005. PMID: 35519060
- As Reacções de Carbonilação e Hidroformilação com Certos Derivados do Indole1. | Shaw, John T. and Floyd T. Tyson. 1956. Journal of the American Chemical Society. 78.11: 2538-2540.
- A síntese do indole de Fischer com ácido fórmico. IA Síntese conveniente de 4a-etil-9-formil-1, 2, 3, 4, 4a, 9a-hexahidro-9H-carbazole. | Ban, Yoshio, et al. 1967. Chemical and Pharmaceutical Bulletin. 15.4: 531-533.
- Dependência do solvente e da temperatura dos espectros de RMN de N-formil e N-tioacetilindolinas. | Nagarajan, K. and M. D. Nair. 1967. Tetrahedron. 23.11: 4493-4497.
- Um estudo espetroscópico de ressonância magnética nuclear de 1H da rotação impedida sobre a ligação amida em alguns N-Formil-2-indolinóis. | LOHSE, CHRISTIAN. 1969. Acta Chemica Scandinavica. 23.4: 1155-1167.
- Síntese de alcalóides de aspidosperma contendo um grupo funcional em C-18; síntese total de (±)-N, O-diacetil-indrocarpinol,(±)-cilindrocarina,(±)-cilindrocarpina,(±)-cilindrocarpidina e (±)-20-alil-20-desetil-20-epiaspidospermina. | Lawton, G., J, et al. 1977. Tetrahedron. 33.13: 1641-1653.
- Conformações e rotação restrita em torno de ligações amida CN de 2, 2-dicloro-1'-formil-3', 4'-dihidrospiro (ciclopropano-1, 2'(1'H)-quinolina) e compostos relacionados. | Suezawa, Hiroko, et al. 1983. Bulletin of the Chemical Society of Japan. 56.5: 1487-1489.
- Formilação de Vilsmeier de tert-anilinas: dibenzo [b, f][1, 5] diazocinas e sais de quinazolínio através do 'efeito t-amino '1. | Cheng, Ying, et al. 1998. Journal of the Chemical Society, Perkin Transactions 1. 7: 1257-1262.
- Síntese de 3-benzilquinoxalina-2 (1H)-onas e 4-formil-3-benzil-3, 4-dihidroquinoxalina-2 (1H)-onas a partir de 3-ariloxirano-2-carboxamidas via 5-arilideno-2, 2-dimetil-1, 3-oxazolidina-4-onas. | Mamedov, Vakhid A., et al. 2022. Tetrahedron. 124: 132963.
Informacoes sobre ordens
| Nome do Produto | Numero de Catalogo | UNID | Preco | Qde | FAVORITOS | |
N-Formylindoline, 25 mg | sc-397086 | 25 mg | $300.00 |
