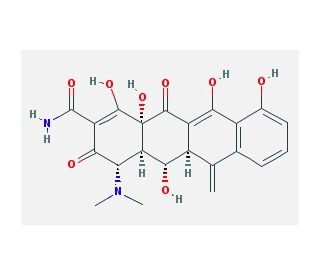
Methacycline (CAS 914-00-1)
QUICK LINKS
Methacycline is a tetracycline antibiotic derivative extensively used in research to investigate bacterial resistance mechanisms and the efficacy of antibiotic regimens. As a broad-spectrum antibiotic, methacycline acts primarily by inhibiting protein synthesis in bacteria, a mechanism it achieves by binding to the 30S ribosomal subunit. This binding disrupts the attachment of aminoacyl-tRNA to the RNA-ribosome complex, thereby inhibiting the addition of new amino acids to the growing peptide chain and effectively halting bacterial growth. In scientific studies, methacycline is particularly valuable for studying the development and transmission of antibiotic resistance. Researchers employ this compound to explore how bacteria evolve resistance to tetracyclines, which involves mutations in ribosomal proteins or changes in the permeability of bacterial cells to the drug. Additionally, methacycline is used in environmental microbiology to study the impact of antibiotic contamination on microbial communities. Its role in influencing microbial dynamics in various ecosystems helps explain broader ecological impacts of antibiotic use and resistance. By understanding these mechanisms, researchers can contribute to the development of more effective strategies for managing antibiotic resistance in clinical and environmental contexts, providing insights that are crucial for the sustainable use of antibiotics.
Methacycline (CAS 914-00-1) References
- Pharmacokinetics and pharmacodynamics of the tetracyclines including glycylcyclines. | Agwuh, KN. and MacGowan, A. 2006. J Antimicrob Chemother. 58: 256-65. PMID: 16816396
- Tetracyclines and pulmonary inflammation. | Rempe, S., et al. 2007. Endocr Metab Immune Disord Drug Targets. 7: 232-6. PMID: 18220943
- Iron supplements: a common cause of drug interactions. | Campbell, NR. and Hasinoff, BB. 1991. Br J Clin Pharmacol. 31: 251-5. PMID: 2054263
- Binding of methacycline to human serum albumin at subdomain IIA using multispectroscopic and molecular modeling methods. | Dong, C., et al. 2013. Luminescence. 28: 933-41. PMID: 23233363
- Inhibition of epithelial-to-mesenchymal transition and pulmonary fibrosis by methacycline. | Xi, Y., et al. 2014. Am J Respir Cell Mol Biol. 50: 51-60. PMID: 23944988
- Methacycline displays a strong efficacy in reducing toxicity in a SCA3 Caenorhabditis elegans model. | Amigoni, L., et al. 2019. Biochim Biophys Acta Gen Subj. 1863: 279-290. PMID: 30420336
- Activity of methacycline, related tetracyclines, and other antibiotics against various L-forms and their parent bacteria in vitro. | Hubert, EG., et al. 1972. Antimicrob Agents Chemother. 2: 276-80. PMID: 4670500
- Demethylchlortetracycline, lymecycline and methacycline compared in patients with purulent exacerbations of bronchitis. | Pines, A., et al. 1968. Br J Dis Chest. 62: 19-26. PMID: 4968585
- Methacycline and demeclocycline in relation to sunlight. | Frost, P., et al. 1971. JAMA. 216: 326-9. PMID: 5107921
- Methacycline compared with ampicillin in acute bacterial exacerbations of chronic bronchitis. A double-blind crossover study. | Chodosh, S., et al. 1976. Chest. 69: 587-92. PMID: 773601
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methacycline, 10 mg | sc-279308 | 10 mg | $133.00 |
