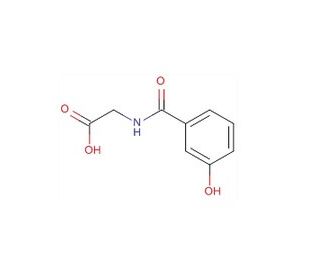
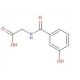
m-Hydroxyhippuric Acid (CAS 1637-75-8)
QUICK LINKS
m-Hydroxyhippuric acid, also known as m-hydroxybenzoylglycine or m-hydroxyhippate, is a type of hippuric acid compound called a hippuric acid. Hippuric acids consist of hippuric acid, which is composed of a glycine attached to a benzoyl group at its N-terminal. m-Hydroxyhippuric acid is found in a solid form, with slight solubility in water, and it exhibits weak acidity based on its pKa value. This acid has been identified in various biofluids, including urine and blood. In the cell, m-hydroxyhippuric acid is primarily located in the cytoplasm. Its biosynthesis occurs from N-benzoylglycine. M-hydroxyhippuric acid, an N-acylglycine, is a metabolite that substitutes the 3rd position on the phenyl ring of hippuric acid (N-benzoylglycine) with a hydroxy group. It functions as both an N-acylglycine and a phenol member. Its origin can be traced back to N-benzoylglycine. It represents the conjugate acid form of m-hydroxyhippurate.
m-Hydroxyhippuric Acid (CAS 1637-75-8) References
- Chocolate intake increases urinary excretion of polyphenol-derived phenolic acids in healthy human subjects. | Rios, LY., et al. 2003. Am J Clin Nutr. 77: 912-8. PMID: 12663291
- Exogenous sources of urinary phenol and indole acids. | SHAW, KN. and TREVARTHEN, J. 1958. Nature. 182: 797-8. PMID: 13590111
- Rennin coagulation of enzymatically dephosphorylated casein. | HSU, R., et al. 1958. Nature. 182: 798-9. PMID: 13590112
- Biological dehydroxylation. | SCHELINE, RR., et al. 1960. Nature. 188: 849-50. PMID: 13747445
- Hippuric acid excretion coffee, and schizophremia. | DASTUR, DK., et al. 1963. Arch Gen Psychiatry. 9: 79-82. PMID: 14025205
- THE RENAL CLEARANCES OF SUBSTITUTED HIPPURIC ACID DERIVATIVES AND OTHER AROMATIC ACIDS IN DOG AND MAN. | Smith, HW., et al. 1945. J Clin Invest. 24: 388-404. PMID: 16695228
- The fate of certain organic acids and amides in the rabbit. 5. o- and m-hydroxybenzoic acids and amides. | Bray, HG., et al. 1948. Biochem J. 43: 561-7. PMID: 16748451
- The fate of certain organic acids and amides in the rabbit. 10. The application of paper chromatography to metabolic studies of hydroxybenzoic acids and amides. | Bray, HG., et al. 1950. Biochem J. 46: 271-5. PMID: 16748674
- Tubular Secretion Markers, Glomerular Filtration Rate, Effective Renal Plasma Flow, and Filtration Fraction in Healthy Adolescents. | Seegmiller, JC., et al. 2020. Kidney Med. 2: 670-672. PMID: 33089146
- Lucerne tannins. 3. Metabolic fate of lucerne tannins in mice. | Milić, BL. and Stojanović, S. 1972. J Sci Food Agric. 23: 1163-7. PMID: 4635999
- Studies on flavonoid metabolism. Absorption and metabolism of (+)-catechin in man. | Das, NP. 1971. Biochem Pharmacol. 20: 3435-45. PMID: 5132890
- Studies on flavonoid metabolism. Metabolism of (+)-[14C] catechin in the rat and guinea pig. | Das, NP. and Griffiths, LA. 1969. Biochem J. 115: 831-6. PMID: 5357023
- Studies on flavonoid metabolism. Metabolism of (+)-catechin in the guinea pig. | Das, NP. and Griffiths, LA. 1968. Biochem J. 110: 449-56. PMID: 5755325
- Measurement of urinary vanilmandelic acid and homovanillic acid by high-performance liquid chromatography with electrochemical detection following extraction by ion-exchange and ion-moderated partition. | Binder, SR. and Sivorinovsky, G. 1984. J Chromatogr. 336: 173-88. PMID: 6526918
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
m-Hydroxyhippuric Acid, 250 mg | sc-490566 | 250 mg | $260.00 | |||
m-Hydroxyhippuric Acid, 2.5 g | sc-490566A | 2.5 g | $2081.00 |
