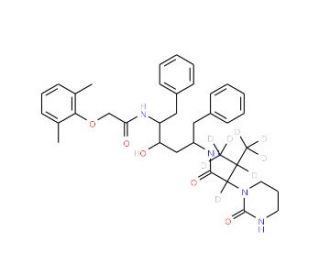
Lopinavir-d8 (CAS 1322625-54-6)
See product citations (1)
QUICK LINKS
Lopinavir-d8 is a deuterated form of lopinavir, where eight hydrogen atoms are replaced by deuterium (heavy hydrogen). This isotopic labeling makes lopinavir-d8 a valuable tool in scientific research for studying the absorption and metabolic pathways of lopinavir without reference to its use in health-related applications. The primary action of lopinavir, and by extension lopinavir-d8, involves the inhibition of HIV-1 protease. This enzyme is essential for the proteolytic cleavage of the viral polyprotein into its functional components, which are necessary for the maturation and assembly of the virus. By binding to the active site of the HIV-1 protease, lopinavir-d8 prevents the enzyme from functioning properly, thereby stopping the virus from maturing and becoming infectious. The deuterated version, lopinavir-d8, retains this mechanism of action and is used in research to track the distribution, metabolism, and excretion of lopinavir with increased precision, thanks to the increased stability and reduced loss of deuterium in mass spectrometry analysis compared to the non-labeled forms. In research, lopinavir-d8 is used extensively in the development and refinement of analytical methods. It serves as an internal standard that provides a consistent reference for quantifying lopinavir in biological samples, aiding in accurate assessments of how lopinavir is processed in various systems. Furthermore, lopinavir-d8 is utilized in studies to determine the detailed pathways through which lopinavir is metabolized by the liver. The presence of deuterium atoms slows the rate of metabolic degradation, allowing for more precise mapping of metabolic pathways. This contributes to identifying the specific enzymes involved in the metabolism of lopinavir and understanding the chemical transformations it undergoes.
Lopinavir-d8 (CAS 1322625-54-6) References
- Validation and application of a high-performance liquid chromatography-tandem mass spectrometric method for simultaneous quantification of lopinavir and ritonavir in human plasma using semi-automated 96-well liquid-liquid extraction. | Wang, PG., et al. 2006. J Chromatogr A. 1130: 302-7. PMID: 16919649
- Validation of an electrospray ionization LC-MS/MS method for quantitative analysis of raltegravir, etravirine, and 9 other antiretroviral agents in human plasma samples. | Quaranta, S., et al. 2009. Ther Drug Monit. 31: 695-702. PMID: 19865000
- Hair and plasma data show that lopinavir, ritonavir, and efavirenz all transfer from mother to infant in utero, but only efavirenz transfers via breastfeeding. | Gandhi, M., et al. 2013. J Acquir Immune Defic Syndr. 63: 578-84. PMID: 24135775
- Determination of total and unbound concentrations of lopinavir in plasma using liquid chromatography-tandem mass spectrometry and ultrafiltration methods. | Illamola, SM., et al. 2014. J Chromatogr B Analyt Technol Biomed Life Sci. 965: 216-23. PMID: 25049210
- Random lopinavir concentrations predict resistance on lopinavir-based antiretroviral therapy. | Court, R., et al. 2016. Int J Antimicrob Agents. 48: 158-62. PMID: 27345268
- Automated high throughput analysis of antiretroviral drugs in dried blood spots. | Duthaler, U., et al. 2017. J Mass Spectrom. 52: 534-542. PMID: 28557187
- Pharmacokinetics of adjusted-dose 8-hourly lopinavir/ritonavir in HIV-infected children co-treated with rifampicin. | Rabie, H., et al. 2019. J Antimicrob Chemother. 74: 2347-2351. PMID: 31081020
- Combination of QSAR, molecular docking, molecular dynamic simulation and MM-PBSA: analogues of lopinavir and favipiravir as potential drug candidates against COVID-19. | Rafi, MO., et al. 2022. J Biomol Struct Dyn. 40: 3711-3730. PMID: 33251975
- Method validation and clinical application for the quantification of lopinavir, efavirenz, and ritonavir in breast milk using liquid chromatography tandem mass spectrometry. | Fraissinet, F., et al. 2022. J Mass Spectrom. 57: e4897. PMID: 36463890
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lopinavir-d8, 1 mg | sc-280927 | 1 mg | $533.00 |
