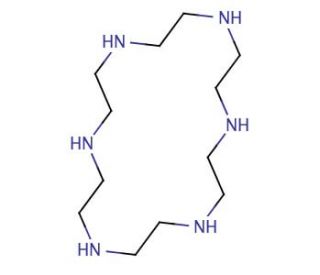
Molecular structure of Hexacyclen, CAS Number: 296-35-5
Hexacyclen (CAS 296-35-5)
Alternate Names:
1,4,7,10,13,16-Hexaazacyclooctadecane
Application:
Hexacyclen is a nitrogen analog of 18-crown-6
CAS Number:
296-35-5
Purity:
≥98%
Molecular Weight:
258.41
Molecular Formula:
C12H30N6
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Hexacyclen is a nitrogen analog of 18-crown-6 (sc-254005), which functions as a good electron donor due to the secondary amines of the molecule, which donate electrons to transition metal cations. Hexacyclen can be used to synthesize ultrathin separation membranes via alternating electrostatic adsorption of Hexacyclen and polyvinylsulfate on porous polyacrylonitrile/polyethylene terephthalate (PAN/PET) substrates. These membranes are very permeable in electrolyte solutions. Chemical studies indicate that Hexacyclen functions as a block copolymer, which is used to synthesize Au(111) nanoplatelets.
Hexacyclen (CAS 296-35-5) References
- Formation and optical properties of gold nanoparticles synthesized in the presence of double-hydrophilic block copolymers. | Yu, SH., et al. 2004. J Nanosci Nanotechnol. 4: 291-8. PMID: 15233092
- Layer-by-layer assembled membranes of protonated 18-azacrown-6 and polyvinylsulfate and their application for highly efficient anion separation. | El-Hashani, A., et al. 2007. J Phys Chem B. 111: 8582-8. PMID: 17411087
- Liquid-liquid extraction of metal ions by the 6-membered N-containing macrocycle hexacyclen. | Arpadjan, S., et al. 1987. Talanta. 34: 953-6. PMID: 18964438
- Polyamine aza-cyclic compounds demonstrate anti-proliferative activity in vitro but fail to control tumour growth in vivo. | Wong, PE., et al. 2010. J Pharm Sci. 99: 4642-57. PMID: 20845462
- The interaction of cations with lipopolysaccharide from Escherichia coli C as shown by measurement of binding constants and aggregation reactions. | Field, AM., et al. 1989. Biochem J. 263: 695-702. PMID: 2688635
- Benchmark Ditopic Binding of Cl- and Cs+ by the Macrocycle Hexacyclen. | Avilés-Moreno, JR., et al. 2017. Chemphyschem. 18: 1324-1332. PMID: 28211620
- Supramolecular reactions of metallo-architectures: Ag2-double-helicate/Zn4-grid, Pb4-grid/Zn4-grid interconversions, and Ag2-double-helicate fusion. | Stadler, AM., et al. 2016. Chem Sci. 7: 3689-3693. PMID: 30008998
- Reversible Multicomponent AND Gate Triggered by Stoichiometric Chemical Pulses Commands the Self-Assembly and Actuation of Catalytic Machinery. | Biswas, PK., et al. 2020. J Am Chem Soc. 142: 7889-7897. PMID: 32286825
- A Switchable Catalyst Duo for Acyl Transfer Proximity Catalysis and Regulation of Substrate Selectivity. | Goswami, A., et al. 2021. Chemistry. 27: 2997-3001. PMID: 33022776
- Using multiple self-sorting for switching functions in discrete multicomponent systems. | Ghosh, A. and Schmittel, M. 2020. Beilstein J Org Chem. 16: 2831-2853. PMID: 33281986
- Structural dynamism of chiral sodium peraza-macrocycle complexes derived from cyclic peptoids. | Schettini, R., et al. 2021. Org Biomol Chem. 19: 7420-7431. PMID: 34397051
- Proton in the ring: spectroscopy and dynamics of proton bonding in macrocycle cavities. | Gámez, F., et al. 2021. Phys Chem Chem Phys. 23: 21532-21543. PMID: 34549205
- Thirteen cationic ionophores: their acute toxicity, neurobehavioral and membrane effects. | Gad, SC., et al. 1985. Drug Chem Toxicol. 8: 451-68. PMID: 4092618
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hexacyclen, 100 mg | sc-255200 | 100 mg | $204.00 |
