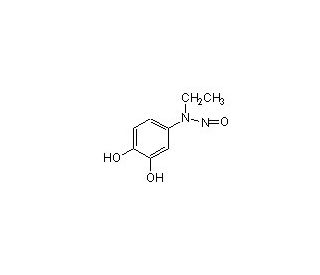

Ethyl-3,4-Dephostatin
Veja citações de produtos (1)
LINKS RÁPIDOS
A etil-3,4-defostatina é um inibidor potente e seletivo conhecido pelo seu intrigante mecanismo de ação, que visa principalmente as proteínas tirosina fosfatases (PTPs). Ao inibir as PTPs, desempenha um papel na modulação das vias de sinalização celular que são fundamentais para vários processos biológicos, incluindo o crescimento celular, a diferenciação e o controlo metabólico. A sua especificidade e eficácia na inibição destas enzimas tornam-na útil na investigação bioquímica, oferecendo conhecimentos sobre os intrincados mecanismos reguladores que regem a função celular. Os investigadores utilizam a etil-3,4-defostatina em aplicações experimentais para elucidar os papéis das PTPs nas vias de sinalização, proporcionando uma compreensão mais profunda da dinâmica celular e contribuindo para o desenvolvimento de novas metodologias de investigação. A sua aplicação no estudo da cinética enzimática e na análise das vias de sinalização exemplifica a sua importância para o avanço no domínio da biologia molecular e da bioquímica.
Ethyl-3,4-Dephostatin Referencias
- Necessidade de actividades da proteína tirosina quinase e da fosfatase para a exocitose do esperma humano. | Tomes, CN., et al. 2004. Dev Biol. 265: 399-415. PMID: 14732401
- Sobrevivência neuronal independente do fator de crescimento nervoso: um papel para os dadores de NO. | Akassoglou, K. 2005. Mol Pharmacol. 68: 952-5. PMID: 16046659
- Inibição da fosfatase 26 de dupla especificidade por etil-3,4-defostatina: Etil-3,4-defostatina como inibidor de multifosfatases. | Seo, H. and Cho, S. 2016. Pharmazie. 71: 196-200. PMID: 27209699
- Identificação de novos inibidores da fosfatase 26 de dupla especificidade através de uma abordagem híbrida de rastreio virtual baseada em farmacóforo e acoplamento molecular. | Ren, JX., et al. 2017. Biomed Pharmacother. 89: 376-385. PMID: 28249240
- Os modificadores de cisteína sugerem um local inibitório alostérico no domínio PDZ da CAL. | Zhao, Y., et al. 2018. Biosci Rep. 38: PMID: 29472314
Informacoes sobre ordens
| Nome do Produto | Numero de Catalogo | UNID | Preco | Qde | FAVORITOS | |
Ethyl-3,4-Dephostatin, 1 mg | sc-220886 | 1 mg | $215.00 |
