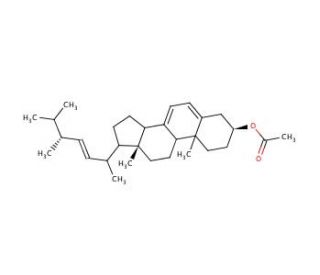
Ergosterol Acetate (CAS 2418-45-3)
QUICK LINKS
Ergosterol acetate, a derivative of the sterol ergosterol found in various organisms, has garnered extensive attention for its potential applications in scientific research. This white, crystalline solid exhibits solubility in organic solvents like ethanol and acetone. Its versatility has been harnessed in diverse laboratory experiments exploring cell membrane structure and enzyme activity. Acting as a substrate for the enzyme sterol acetyltransferase (SAT), ergosterol acetate plays a role in the synthesis of cholesterol from ergosterol. The enzyme catalyzes the transfer of an acetyl group from acetyl-CoA to the sterol molecule, leading to cholesterol formation, subsequently incorporating into cell membranes. Moreover, ergosterol acetate can also function as an enzyme inhibitor, facilitating the study of its activity.
Ergosterol Acetate (CAS 2418-45-3) References
- Fungicidal properties of meso-arylglycosylporphyrins: influence of sugar substituents on photoinduced damage in the yeast Saccharomyces cerevisiae. | Carré, V., et al. 1999. J Photochem Photobiol B. 48: 57-62. PMID: 10205879
- Small-molecule diversity using a skeletal transformation strategy. | Kumar, N., et al. 2005. Org Lett. 7: 2535-8. PMID: 15957884
- New photoantimicrobial films composed of porphyrinated lipophilic cellulose esters. | Krouit, M., et al. 2006. Bioorg Med Chem Lett. 16: 1651-5. PMID: 16377183
- Photobactericidal plastic films based on cellulose esterified by chloroacetate and a cationic porphyrin. | Krouit, M., et al. 2008. Bioorg Med Chem. 16: 10091-7. PMID: 18954991
- Nitroso Diels-Alder (NDA) reaction as an efficient tool for the functionalization of diene-containing natural products. | Carosso, S. and Miller, MJ. 2014. Org Biomol Chem. 12: 7445-68. PMID: 25119424
- Phytosterols Suppress Phagocytosis and Inhibit Inflammatory Mediators via ERK Pathway on LPS-Triggered Inflammatory Responses in RAW264.7 Macrophages and the Correlation with Their Structure. | Yuan, L., et al. 2019. Foods. 8: PMID: 31744147
- Characterization of a Pentacyclic Triterpene Acetyltransferase Involved in the Biosynthesis of Taraxasterol and ψ-Taraxasterol Acetates in Lettuce. | Choi, HS., et al. 2021. Front Plant Sci. 12: 788356. PMID: 35046976
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ergosterol Acetate, 100 mg | sc-218327 | 100 mg | $405.00 |
