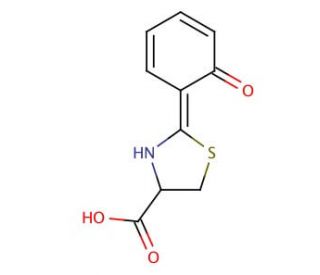
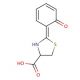
Dihydroaeruginoic acid の分子構造、CAS番号: 143209-04-5
Dihydroaeruginoic acid (CAS 143209-04-5)
アプリケーション:
Dihydroaeruginoic acidは単純な芳香族シデロフォアである
CAS 番号:
143209-04-5
分子量:
223.25
分子式:
C10H9NO3S
試験・研究用以外には使用しないでください。 臨床及び体外診断には使用できません。
* Refer to Certificate of Analysis for lot specific data.
クイックリンク
注文情報
説明
技術サポート情報
安全情報
SDSと分析証明書
ジヒドロアエルギン酸 (DHAA) は,数種のPseudomonas,植物および菌類によって産生される単純な芳香族シデロホアである。それはその生化学的および生理学的効果を研究するために多くの実験室実験で使用されてきた。抗菌、抗真菌、抗ウイルス、抗酸化および抗腫瘍活性を示す。DHAAは、炭水化物、タンパク質、脂質の代謝に関与する酵素の阻害を介して作用する。また,活性酸素種を除去し,酸化ストレスを低減する。さらに,細胞周期調節,アポトーシスおよび細胞移動に関与する遺伝子の発現を調節することが示されている。
Dihydroaeruginoic acid (CAS 143209-04-5) 参考文献
- マイコバクテリアにおける鉄制御遺伝子の比較解析。 | Yellaboina, S., et al. 2006. FEBS Lett. 580: 2567-76. PMID: 16631750
- 海洋放線菌Salinispora tropica CNB-440の可変ヒドロキサム酸シデロフォア代謝系。 | Ejje, N., et al. 2013. Metallomics. 5: 1519-28. PMID: 24121533
- トランスポゾン指向性挿入部位配列決定によるBurkholderia thailandensisの土壌生存に必要な遺伝子の同定。 | Bishop, AH. and Rachwal, PA. 2014. Curr Microbiol. 68: 693-701. PMID: 24488501
- Pseudomonas fluorescens G308が生産するN-メルカプト-4-ホルミルカルボスチリルから2-(2-ヒドロキシフェニル)チアゾール-4-カルバルデヒド[aeruginaldehyde]への構造変化。 | Ye, L., et al. 2014. Nat Prod Commun. 9: 789-94. PMID: 25115080
- シュードモナス・プロテジェンスPf-5のエナンチオピオケリン生合成遺伝子クラスターに関連するトランスポーターの破壊は多面的な影響を及ぼす。 | Lim, CK., et al. 2016. PLoS One. 11: e0159884. PMID: 27442435
- 細菌の一対のシデロフォアが細胞間シグナル分子を放出し, 捕捉する:天然ニトロンのバイオコンジュゲーションの珍しい例。 | Trottmann, F., et al. 2019. Angew Chem Int Ed Engl. 58: 200-204. PMID: 30375753
- 緑膿菌IQS論争に終止符を打つ。 | Cornelis, P. 2020. Microbiologyopen. 9: e962. PMID: 31667921
- 緑膿菌が分泌する複数の化合物が, 黄色ブドウ球菌の抗菌性金属である銅と銀に対する耐性を高める。 | Monych, NK. and Turner, RJ. 2020. mSystems. 5: PMID: 32900873
- バークホルデリア菌がカルバペネム耐性グラム陰性細菌病原体を阻害する複数の新規可能性分子を産生。 | Depoorter, E., et al. 2021. Antibiotics (Basel). 10: PMID: 33540653
- クロスメタボロームアプローチにより, コムギは根の代謝産物を通じて蛍光性シュードモナスの生理を阻害することが明らかになった。 | Rieusset, L., et al. 2021. Metabolites. 11: PMID: 33572622
- ピオケリン生合成代謝物が鉄を結合し, シデロフォア様活性を示すシュードモナドの成長を促進する。 | Kaplan, AR., et al. 2021. ACS Infect Dis. 7: 544-551. PMID: 33577297
- 緑膿菌におけるピロヘリンとジヒドロエルギノ酸の生合成には, 鉄を制御するpchDCBAオペロンが必要である。 | Serino, L., et al. 1997. J Bacteriol. 179: 248-57. PMID: 8982005
- ジヒドロエルギノ酸類似体のハイスループット合成へのアプローチ | Loughlin, W. A., Knevitt, S. A., Hosking, R. E., & Marshall, R. L. 2000. Australian Journal of Chemistry. 53(6): 457-462.
注文情報
| 製品名 | カタログ # | 単位 | 価格 | 数量 | お気に入り | |
Dihydroaeruginoic acid, 5 mg | sc-391518 | 5 mg | $194.00 |
