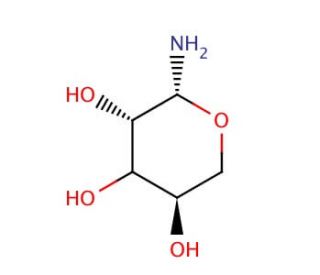
D-Ribopyranosylamine (CAS 43179-09-5)
QUICK LINKS
D-Ribopyranosylamine, a derivative of ribose, has emerged as a pivotal compound in biochemical and pharmaceutical research due to its role as a precursor in the synthesis of various bioactive molecules and glycoconjugates. In research, this chemical serves as a key intermediate in the production of nucleoside analogs, glycosylation reactions, and the modification of nucleic acids and carbohydrates. Its chemical structure, comprising a ribose sugar moiety and an amino group, facilitates its incorporation into nucleotide analogs, where it can mimic the natural nucleosides, thereby influencing nucleic acid structure and function. Additionally, D-Ribopyranosylamine is utilized in the enzymatic synthesis of glycosidic bonds, where it acts as a glycosyl donor or acceptor in glycosylation reactions mediated by glycosyltransferases. Moreover, researchers exploit its reactivity and functional group compatibility to introduce diverse chemical modifications into carbohydrates and nucleic acids, enabling the generation of structurally diverse compounds with potential biological activities. Furthermore, D-Ribopyranosylamine derivatives have shown promise in medicinal chemistry research, particularly in the development of nucleoside-based antiviral, anticancer, and antimicrobial agents. Through its versatile applications in chemical synthesis and biological research, D-Ribopyranosylamine continues to contribute to advancements in understanding carbohydrate metabolism, nucleic acid biochemistry, and drug discovery efforts.
D-Ribopyranosylamine (CAS 43179-09-5) References
- Purines, pyrimidines, and imidazoles. XL. A new synthesis of a D-ribofuranosylamine derivative and its use in the synthesis of pyrimidine and imidazole nucleosides. | Cusack, NJ., et al. 1973. J Chem Soc Perkin 1. 16: 1720-31. PMID: 4796433
- Purines, pyrimidines, and imidazoles. Part 46. Some acyclic D-arabinose imidazole and purine nucleosides. | Mackenzie, G., et al. 1977. J Chem Soc Perkin 1. 1094-1103. PMID: 559007
- A simple synthesis in high yield of 2,3-O-isopropylidene-β-D-ribofuranosylamine, an intermediate in the preparation of N-substituted ribofuranosides | Cusack, N. J., & Shaw, G. 1970. Journal of the Chemical Society D: Chemical Communications. 17: 1114-1114.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
D-Ribopyranosylamine, 1 g | sc-207481A | 1 g | $290.00 | |||
D-Ribopyranosylamine, 2.5 g | sc-207481 | 2.5 g | $390.00 | |||
D-Ribopyranosylamine, 5 g | sc-207481B | 5 g | $650.00 | |||
D-Ribopyranosylamine, 10 g | sc-207481C | 10 g | $1240.00 | |||
D-Ribopyranosylamine, 25 g | sc-207481D | 25 g | $2800.00 |
