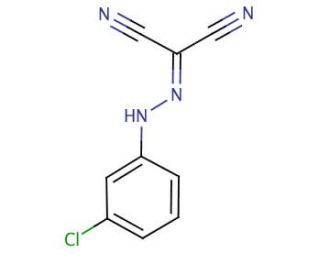
Carbonyl Cyanide m-Chlorophenylhydrazone 의 분자 구조, CAS 번호: 555-60-2
Carbonyl Cyanide m-Chlorophenylhydrazone (CAS 555-60-2)
인용논문 보기 (8)
대체 이름:
(3-Chlorophenyl)hydrazonomalononitrile; Mesoxalonitrile 3-chlorophenylhydrazone; CCCP
적용:
Carbonyl Cyanide m-Chlorophenylhydrazone 는 광합성의 결합 해제제 및 억제제로 일반적으로 사용되는 양성자 이오노포어입니다.
CAS 등록번호:
555-60-2
순도:
≥97%
분자량:
204.62
분자식:
C9H5ClN4
추가 정보:
이는 운송용 위험물이며 추가 배송료가 부과될 수 있습니다.
연구용으로만 사용가능합니다. 진단이나 치료용으로 사용불가합니다.
* 참조분석증명서대량의 측정 데이터(함수량포함).
빠른 링크
주문정보
논문정보
설명
기술정보
안전정보
SDS 및 분석 증명서
CCCP(Carbonyl Cyanide m-Chloropenylhydrazone)는 다재다능한 합성 화합물로 과학 연구에 광범위하게 사용된다. 무색의 고체로 존재하며 극성 유기 용매에 용해되며, 약물 합성의 중간체로서 합성 유기 화학 및 의약 화학 분야에서 광범위한 응용을 발견한다. CCCP는 프로스타글란딘 합성에 관여하는 효소 억제, 사이클로옥시게나제 활성 억제 등 다양한 생화학적 및 생리학적 효과를 나타낸다. 항염증성, 진통성, 항종양성 등을 입증하였다. 또한 CCCP는 약물, 세포 및 조직에 미치는 영향을 조사하는 등 다양한 과학 연구 분야에 활용되고 있다. 프로스타글란딘 합성 효소를 저해하고 프로스타글란딘 생성에 중요한 사이클로옥시게나제를 경쟁적으로 저해하는 것으로 알려져 있다. 또한 CCCP는 프로톤 이온 전달체로서 미토콘드리아 막 및 엽록체 막에 대한 효과로 인해 분리제 및 광합성 억제제로서 일반적으로 사용된다. ER-Golgi 단백질 수송 억제제로서 시토크롬 C 산화효소와의 결합성이 높다. 황소개구리 교감신경절 실험에서 CCCP는 루테인화 호르몬 방출 호르몬의 자발적인 방출을 증가시켰고, 발화 빈도에 크게 의존했다. 또한 CCCP가 양성자 전도체로서 작용하여 포도당 존재하에서 대장균 세포의 생존성에 영향을 미칠 수 있다는 연구 결과가 있다.
Carbonyl Cyanide m-Chlorophenylhydrazone (CAS 555-60-2) 참고자료
- 카페인과 카보닐 시안화물 m-클로로페닐히드라존은 온전한 시냅스 전 단자에서 황체 형성 호르몬 방출 호르몬의 유발 및 자발적 방출을 증가시켰습니다. | Cao, YJ. and Peng, YY. 1999. Neuroscience. 92: 1511-21. PMID: 10426503
- HL-60에서 카스파제-3 활성 및 카보닐 시안화물 m-클로로페닐히드라존에 의한 세포 사멸. | Mlejnek, P. 2001. Altern Lab Anim. 29: 243-9. PMID: 11387021
- 효모 사카로미세스 세레비지애에서 칼슘 신호와 혈장막 H(+)-ATPase의 활성화를 유도하는 시안화 메클로로페닐히드라존. | Pereira, MB., et al. 2008. FEMS Yeast Res. 8: 622-30. PMID: 18399987
- 미토콘드리아 언커플러 카르보닐 시아나이드 M-클로로페닐히드라존은 복구 효소 단백질 L-소아스파르틸 메틸 트랜스퍼라제의 멀티머 조립 및 활성을 유도합니다. | Fanélus, I. and Desrosiers, RR. 2013. J Mol Neurosci. 50: 411-23. PMID: 23319267
- 시안화 카르보닐 m-클로로페닐히드라존이 대장균 할로토로내성에 미치는 영향. | Ghoul, M., et al. 1989. Appl Environ Microbiol. 55: 1040-3. PMID: 2658803
- 미토콘드리아 언커플러 카르보닐 시아나이드 m-클로로페닐히드라존은 동맥 평활근 세포에서 KATP 채널 활성화와 관련 없이 혈관 이완을 유도합니다. | Zhang, YQ., et al. 2016. Br J Pharmacol. 173: 3145-3158. PMID: 27534899
- 개구리 운동 신경 단자의 정량적 전달 물질 방출 및 초구조에 대한 시안화 카르보닐 m-클로로페닐히드라존의 효과. | Molgo, J. and Pecot-Dechavassine, M. 1988. Neuroscience. 24: 695-708. PMID: 2834667
- 모체 노화는 젖소의 카보닐 시안화물-m-클로로페닐히드라존에 의한 미토콘드리아 기능 장애에 대한 난모세포 회복력에 영향을 미칩니다. | Kansaku, K., et al. 2017. PLoS One. 12: e0188099. PMID: 29182624
- 산화 인산화 카르보닐 시아나이드 m-클로로페닐히드라존의 고전적인 언커플러의 미토콘드리아 표적 유도체는 효과적인 미토콘드리아 리커플러입니다. | Iaubasarova, IR., et al. 2020. PLoS One. 15: e0244499. PMID: 33378414
- 시안화탄소 m-클로로페닐히드라존이 대장균의 호흡 및 호흡 의존적 인산화에 미치는 영향. | Cavari, BZ. and Avi-Dor, Y. 1967. Biochem J. 103: 601-8. PMID: 4962086
- 시안화탄소 m-클로로페닐히드라존이 대장균과 할로토내성 브레비박테리움 종에 미치는 치명적인 영향. | Nagata, S. 1995. Microbios. 81: 73-83. PMID: 7476556
- 카르보닐 시안화물 3-클로로페닐히드라존과 시토크롬 C 산화효소의 상호작용. | Bona, M., et al. 1993. Gen Physiol Biophys. 12: 533-42. PMID: 8070645
- 시안화 카르보닐 m-클로로페닐히드라존과 광합성 II 수용체 측의 상호 작용. | Samuilov, VD. and Barsky, EL. 1993. FEBS Lett. 320: 118-20. PMID: 8458426
- 카르보닐 시안화물 m-클로로페닐히드라존(CCCP)이 지단백질 리파아제의 이량체화에 미치는 영향. | Park, JW., et al. 1997. Biochim Biophys Acta. 1344: 132-8. PMID: 9030190
의 억제제:
1810026J23Rik, 1810043H04Rik, 4930420K17Rik, AFG3L1, AFG3L2, apoOL, ATAD3B, BRP44L, C7orf44, C7orf73, C8orf38, CCDC58, Cephalosporin, CHCHD8, CHPPR, Clpb, Cstad, Cytokeratin 14, D10Wsu102e, E. coli K99, EG546368, EG623286, ERAL1, FASTKD3, FDX1L, Francisella tularensis LPS, GDAP1, GDAP1L1, GTPBP3, HIGD1B, HIGD2A, IARS2, ICT1, IMMP1L, IMMP2L, IMPA1, KLH-B, LACE1, LONP2, LYCAT, MCART2, MINOS1, MIPEP, Misato, Mitochondrial Marker, Mitofilin, MPV17, MPV17L2, MTERFD1, MTMR12, mtp110, MTRF1, NDUFB9, NIMP, Nix, Olfr631, OPA1, OSAP, OXA1L, P117, Paraplegin, Parkin, PINK1, PMCA1, PMPCB, PTCD3, QIL1, Rho T2, RPUSD2, Salmonella flagella, Sam50, SFXN3, SFXN4, SLC25A35, SLMO2, SMCR7L, Spatacsin, Syntaphilin, Syntaxin 7, Tim13A/B, Tim17B, Tim22, Tim50, TMEM11, TMEM126A, Tom22, Tom34, Tom7, Tom70, Tomm5, Twinkle, UQCRH, UT-B, YME1L1.의 활성화자:
4930420K17Rik, Apaf-1, ATP6L, ATP8, ATP8B5, C12orf65_2810006K23Rik, CALCOCO1, ClpP, COX8a, Cstad, DRP1, Fis1, FUNDC1, GHITM, HCCR-1, LONP2, MCJ, MIOS, Mitoferrin, MRP-L20, MTCH1, MTFmt, NDUFV3, NOD9, OPA3, OTOP2, PARL, RAIDD, SFXN1, SLC25A18, SLC25A22, SLC25A40, SLC25A45, SLMAP, Tim23, Tom20, Tomm20l, V-ATPase D2, VDAC2.주문정보
| 제품명 | 카탈로그 번호 | 단위 | 가격 | 수량 | 관심품목 | |
Carbonyl Cyanide m-Chlorophenylhydrazone, 100 mg | sc-202984A | 100 mg | $77.00 | |||
Carbonyl Cyanide m-Chlorophenylhydrazone, 250 mg | sc-202984 | 250 mg | $153.00 | |||
Carbonyl Cyanide m-Chlorophenylhydrazone, 500 mg | sc-202984B | 500 mg | $240.00 |
