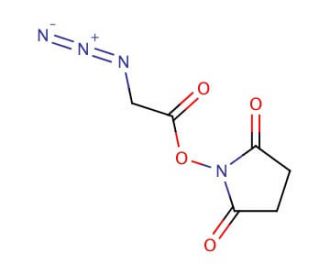
Azidoacetic acid NHS ester (CAS 824426-32-6)
QUICK LINKS
Azidoacetic acid NHS ester is a chemical compound that acts as a versatile and reactive moiety in the field of bioconjugation chemistry, particularly within the context of click chemistry reactions. This compound is characterized by its azido group, which enables it to participate in highly selective and efficient copper(I)-catalyzed azide-alkyne cycloaddition (CuAAC) reactions, also known as "click" reactions. The presence of the N-hydroxysuccinimide (NHS) ester group further enhances its utility by facilitating the formation of stable amide bonds with primary amines found on biomolecules, such as proteins or nucleic acids. This dual functionality makes Azidoacetic acid NHS ester an invaluable tool in the synthesis of complex molecules for research, allowing for the introduction of azide functional groups into biomolecules in a controlled manner. This is for the development of labeled compounds, probes, or crosslinked complexes in the study of molecular interactions, cellular processes, and the elucidation of biological pathways without perturbing the native biological systems. The specificity and efficiency of the reactions involving Azidoacetic acid NHS ester have positioned it as a cornerstone reagent in the toolbox of modern molecular biology and synthetic chemistry research.
Azidoacetic acid NHS ester (CAS 824426-32-6) References
- Chemoenzymatic synthesis of CMP-sialic acid derivatives by a one-pot two-enzyme system: comparison of substrate flexibility of three microbial CMP-sialic acid synthetases. | Yu, H., et al. 2004. Bioorg Med Chem. 12: 6427-35. PMID: 15556760
- Immobilization of carbohydrate epitopes for surface plasmon resonance using the Staudinger ligation. | Loka, RS. and Cairo, CW. 2010. Carbohydr Res. 345: 2641-7. PMID: 20971453
- Functionalizable Amine-based Polymer Nanoparticles. | Wang, H., et al. 2013. ACS Macro Lett. 2: 948-951. PMID: 25580372
- Characterizing non-hydrolyzing Neisseria meningitidis serogroup A UDP-N-acetylglucosamine (UDP-GlcNAc) 2-epimerase using UDP-N-acetylmannosamine (UDP-ManNAc) and derivatives. | Zhang, L., et al. 2016. Carbohydr Res. 419: 18-28. PMID: 26598987
- Conjugation and Evaluation of Triazole-Linked Single Guide RNA for CRISPR-Cas9 Gene Editing. | He, K., et al. 2016. Chembiochem. 17: 1809-1812. PMID: 27441384
- Antigen-Drug Conjugates as a Novel Therapeutic Class for the Treatment of Antigen-Specific Autoimmune Disorders. | Pickens, CJ., et al. 2019. Mol Pharm. 16: 2452-2461. PMID: 31083955
- Site-Specific Modification of Proteins through N-Terminal Azide Labeling and a Chelation-Assisted CuAAC Reaction. | Inoue, N., et al. 2019. Bioconjug Chem. 30: 2427-2434. PMID: 31436410
- Characterization of Brain Inflammation, Apoptosis, Hypoxia, Blood-Brain Barrier Integrity and Metabolism in Venezuelan Equine Encephalitis Virus (VEEV TC-83) Exposed Mice by In Vivo Positron Emission Tomography Imaging. | Bocan, TM., et al. 2019. Viruses. 11: PMID: 31766138
- An efficient gene knock-in strategy using 5'-modified double-stranded DNA donors with short homology arms. | Yu, Y., et al. 2020. Nat Chem Biol. 16: 387-390. PMID: 31873222
- c(RGDfK)- and ZnTriMPyP-Bound Polymeric Nanocarriers for Tumor-Targeted Photodynamic Therapy. | de Las Heras, E., et al. 2020. Photochem Photobiol. 96: 570-580. PMID: 32104926
- The Minimal Effect of Linker Length for Fatty Acid Conjugation to a Small Protein on the Serum Half-Life Extension. | Cho, J., et al. 2020. Biomedicines. 8: PMID: 32357510
- Supramolecular polymerization of sulfated dendritic peptide amphiphiles into multivalent L-selectin binders. | Straßburger, D., et al. 2021. Beilstein J Org Chem. 17: 97-104. PMID: 33519996
- Nanoenzyme engineered neutrophil-derived exosomes attenuate joint injury in advanced rheumatoid arthritis via regulating inflammatory environment. | Zhang, L., et al. 2022. Bioact Mater. 18: 1-14. PMID: 35387158
- Photoactivatable Circular Caged Oligonucleotides for Transcriptome In Vivo Analysis (TIVA). | Yang, L., et al. 2021. ChemPhotoChem. 5: 940-946. PMID: 35434268
- Strategies for the Biofunctionalization of Straining Flow Spinning Regenerated Bombyx mori Fibers. | Lozano-Picazo, P., et al. 2022. Molecules. 27: PMID: 35807389
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Azidoacetic acid NHS ester, 100 mg | sc-496405 | 100 mg | $208.00 |
