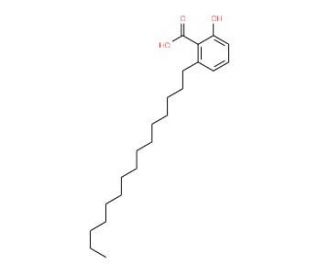
Anacardic Acid (CAS 16611-84-0)
See product citations (13)
QUICK LINKS
Anacardic Acid is a pentadecane aliphatic chain containing hydroxylcarboxylic acid. The acid is an effective inhibitor of the activities of p300, p300/CBP associated factor histone acetyltransferase, prostaglandin synthase (PTGS2 or Cox-2), tyrosinase, urease-α and -β, lipoxygenase (LO), and xanthine oxidase. Additionally, it may act to potently inhibit HAT-dependent transcription and SUMOylation, as well as decrease expression of NF-kappaB regulated gene products at high concentrations. Research suggests that anacardic acid is a potent activator of ARK-1 (Aurora kinase A) mediated phosphorylation of histone H3 while leaving ARK-2 unaffected in vitro. Anacardic acid has demonstrated bactericidal activity against methicillin-resistant Staphylococcus aureus (MRSA).
Anacardic Acid (CAS 16611-84-0) References
- Synthesis of sildenafil analogues from anacardic acid and their phosphodiesterase-5 inhibition. | Paramashivappa, R., et al. 2002. J Agric Food Chem. 50: 7709-13. PMID: 12475293
- Small molecule modulators of histone acetyltransferase p300. | Balasubramanyam, K., et al. 2003. J Biol Chem. 278: 19134-40. PMID: 12624111
- Inhibition of histone acetyltransferase activity by anacardic acid sensitizes tumor cells to ionizing radiation. | Sun, Y., et al. 2006. FEBS Lett. 580: 4353-6. PMID: 16844118
- Specific small-molecule activator of Aurora kinase A induces autophosphorylation in a cell-free system. | Kishore, AH., et al. 2008. J Med Chem. 51: 792-7. PMID: 18215015
- Anacardic acid (6-nonadecyl salicylic acid), an inhibitor of histone acetyltransferase, suppresses expression of nuclear factor-kappaB-regulated gene products involved in cell survival, proliferation, invasion, and inflammation through inhibition of the inhibitory subunit of nuclear factor-kappaBalpha kinase, leading to potentiation of apoptosis. | Sung, B., et al. 2008. Blood. 111: 4880-91. PMID: 18349320
- Histone acetyltransferase inhibitor anacardic acid causes changes in global gene expression during in vitro Plasmodium falciparum development. | Cui, L., et al. 2008. Eukaryot Cell. 7: 1200-10. PMID: 18487348
- Ginkgolic acid inhibits protein SUMOylation by blocking formation of the E1-SUMO intermediate. | Fukuda, I., et al. 2009. Chem Biol. 16: 133-40. PMID: 19246003
- Anacardic acid induces caspase-independent apoptosis and radiosensitizes pituitary adenoma cells. | Sukumari-Ramesh, S., et al. 2011. J Neurosurg. 114: 1681-90. PMID: 21275565
- Anacardic acid inhibits the catalytic activity of matrix metalloproteinase-2 and matrix metalloproteinase-9. | Omanakuttan, A., et al. 2012. Mol Pharmacol. 82: 614-22. PMID: 22745359
- Anacardic acid (6-pentadecylsalicylic acid) induces apoptosis of prostate cancer cells through inhibition of androgen receptor and activation of p53 signaling. | Tan, J., et al. 2012. Chin J Cancer Res. 24: 275-83. PMID: 23359208
- Induction of the endoplasmic reticulum stress and autophagy in human lung carcinoma A549 cells by anacardic acid. | Seong, YA., et al. 2014. Cell Biochem Biophys. 68: 369-77. PMID: 23955513
- Anacardic acid induces cell apoptosis of prostatic cancer through autophagy by ER stress/DAPK3/Akt signaling pathway. | Tan, J., et al. 2017. Oncol Rep. 38: 1373-1382. PMID: 28731173
- Transcriptomic response of breast cancer cells to anacardic acid. | Schultz, DJ., et al. 2018. Sci Rep. 8: 8063. PMID: 29795261
- Ginkgolic acid and anacardic acid are specific covalent inhibitors of SARS-CoV-2 cysteine proteases. | Chen, Z., et al. 2021. Cell Biosci. 11: 45. PMID: 33640032
- Antibacterial activity of anacardic acid and totarol, alone and in combination with methicillin, against methicillin-resistant Staphylococcus aureus. | Muroi, H. and Kubo, I. 1996. J Appl Bacteriol. 80: 387-94. PMID: 8849640
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Anacardic Acid, 5 mg | sc-202463 | 5 mg | $102.00 | |||
Anacardic Acid, 25 mg | sc-202463A | 25 mg | $204.00 |
