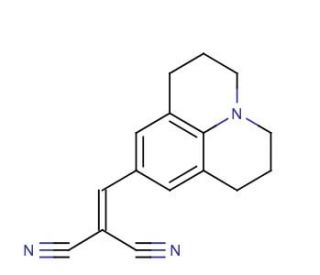
9-(2,2-Dicyanovinyl)julolidine (CAS 58293-56-4)
LINKS RÁPIDOS
A 9-(2,2-Dicyanovinyl)julolidine, referida como DCVJ, é um composto orgânico sintético amplamente utilizado em diversos projectos de investigação científica. Esta molécula altamente reactiva e versátil é a escolha ideal para um vasto espetro de experiências laboratoriais. Apresenta solubilidade em água, álcool e éter. As aplicações da 9-(2,2-Dicyanovinyl)julolidine na investigação científica abrangem uma vasta gama. Serve como um reagente valioso na síntese orgânica, um catalisador em reacções de polimerização e um solvente na preparação de polímeros e vários materiais. Funcionando como um nucleófilo, a 9-(2,2-dicianovinil)julolidina possui a capacidade de formar ligações covalentes com espécies deficientes em electrões. Esta reação ocorre quando o nucleófilo ataca a espécie deficiente em electrões, como um alceno ou um grupo carbonilo, resultando no estabelecimento de uma nova ligação covalente. Esta reação específica, conhecida como reação de adição, serve de base a várias reacções orgânicas sintéticas.
9-(2,2-Dicyanovinyl)julolidine (CAS 58293-56-4) Referencias
- Anisotropia de fluorescência de rotores moleculares. | Levitt, JA., et al. 2011. Chemphyschem. 12: 662-72. PMID: 21328515
- Dependência intrínseca e extrínseca da temperatura de rotores moleculares fluorescentes sensíveis à viscosidade. | Howell, S., et al. 2012. J Fluoresc. 22: 457-65. PMID: 21947609
- Uma abordagem baseada na citometria de fluxo para facilitar a quantificação, a estimativa do tamanho e a caraterização de partículas subvisíveis em soluções proteicas. | Lubich, C., et al. 2015. Pharm Res. 32: 2863-76. PMID: 25788448
- Não tocar! A abrasão da proteína adsorvida é a causa principal da formação de partículas subvisíveis durante a agitação. | Sediq, AS., et al. 2016. J Pharm Sci. 105: 519-529. PMID: 26869415
- Deteção de ligação por fluorescência da formação de duplex de ADN por um análogo tricíclico da citidina. | Burns, DD., et al. 2017. J Am Chem Soc. 139: 1372-1375. PMID: 28080035
- Estimulação da libertação de glutamato do sinaptoneurossoma pela α-sinucleína monomérica e fibrilada. | Sarafian, TA., et al. 2017. J Neurosci Res. 95: 1871-1887. PMID: 28117497
- Fluorescência dependente do fluxo da CCVJ. | Schmidt, MJ., et al. 2017. J Biol Eng. 11: 24. PMID: 28785307
- As interacções electrostáticas lípido-proteína sequestram a dobra amiloide curli na superfície da membrana lipopolissacarídica. | Swasthi, HM. and Mukhopadhyay, S. 2017. J Biol Chem. 292: 19861-19872. PMID: 29021250
- Adaptabilidade composicional em redes de andaimes NPM1-SURF6 possibilitada pela comutação dinâmica de mecanismos de separação de fases. | Ferrolino, MC., et al. 2018. Nat Commun. 9: 5064. PMID: 30498217
- Compostos à base de colesterol: Avanços recentes em síntese e aplicações. | Albuquerque, HMT., et al. 2018. Molecules. 24: PMID: 30597999
- Um Método para Medições de Alto Rendimento da Viscosidade em Sistemas de Membranas de Dimensão Sub-micrométrica. | Chwastek, G., et al. 2020. Chembiochem. 21: 836-844. PMID: 31566864
- Deteção e quantificação de nanopartículas de poliestireno não marcadas utilizando um rotor molecular fluorescente. | Moraz, A. and Breider, F. 2021. Anal Chem. 93: 14976-14984. PMID: 34735123
- Isolamento e Quantificação de Nanoplásticos de Poliestireno em Tecidos por Cromatografia de Exclusão de Tamanho a Baixa Pressão. | Gagné, F. 2022. J Xenobiot. 12: 109-121. PMID: 35645291
Informacoes sobre ordens
| Nome do Produto | Numero de Catalogo | UNID | Preco | Qde | FAVORITOS | |
9-(2,2-Dicyanovinyl)julolidine, 5 mg | sc-210694 | 5 mg | $52.00 |
