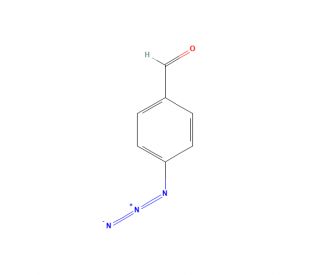
Molecular structure of 4-Azidobenzaldehyde, CAS Number: 24173-36-2
4-Azidobenzaldehyde (CAS 24173-36-2)
See product citations (1)
Alternate Names:
p-Azidobenzaldehyde; p-Azido-benzaldehyde
CAS Number:
24173-36-2
Molecular Weight:
147.13
Molecular Formula:
C7H5N3O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Azidobenzaldehyde is classified as a nitrobenzaldehyde and is used in organic synthesis due to its reactivity. Its reactivity is due to the presence of the azide group, which can be used to form a variety of covalent bonds with other molecules. The azide group can also be used to form a variety of different types of bonds with other molecules, such as carbon-carbon bonds, carbon-nitrogen bonds, and carbon-oxygen bonds. Additionally, It may act to inhibit the enzyme cytochrome P450. It is used as a reagent in organic synthesis, a starting material for the production of pharmaceuticals, a substrate in enzyme assays, and a fluorescent label for biological molecules.
4-Azidobenzaldehyde (CAS 24173-36-2) References
- p-Phenylenecarbenonitrene and its halogen derivatives: how does resonance interaction between a nitrene and a carbene center affect the overall electronic configuration? | Nicolaides, A., et al. 2001. J Am Chem Soc. 123: 2628-36. PMID: 11456932
- Photolysis and oxidation of azidophenyl-substituted radicals: delocalization in heteroatom-based radicals. | Serwinski, PR., et al. 2004. J Org Chem. 69: 5247-60. PMID: 15287767
- Azido push-pull fluorogens photoactivate to produce bright fluorescent labels. | Lord, SJ., et al. 2010. J Phys Chem B. 114: 14157-67. PMID: 19860443
- Copper(II)-Catalyzed Conversion of Aryl/Heteroaryl Boronic Acids, Boronates, and Trifluoroborates into the Corresponding Azides: Substrate Scope and Limitations. | Grimes, KD., et al. 2010. Synthesis (Stuttg). 2010: 1441-1448. PMID: 20526454
- Chemoselective reduction of the carbonyl functionality through hydrosilylation: integrating click catalysis with hydrosilylation in one pot. | Roy, SR., et al. 2014. J Org Chem. 79: 9150-60. PMID: 25188382
- Easy access to heterobimetallic complexes for medical imaging applications via microwave-enhanced cycloaddition. | Desbois, N., et al. 2015. Beilstein J Org Chem. 11: 2202-8. PMID: 26664643
- Synthesis and Biochemical Evaluation of 3-Phenoxy-1,4-diarylazetidin-2-ones as Tubulin-Targeting Antitumor Agents. | Greene, TF., et al. 2016. J Med Chem. 59: 90-113. PMID: 26680364
- Purine-Type Compounds Induce Microtubule Fragmentation and Lung Cancer Cell Death through Interaction with Katanin. | Kuo, TC., et al. 2016. J Med Chem. 59: 8521-34. PMID: 27536893
- pH-Responsive Nanoparticles Based on Covalently Grafted Conjugates of Carboxymethyl Chitosan and Daunorubicin for the Delivery of Anti-Cancer Drugs. | Cao, J., et al. 2017. J Biomed Nanotechnol. 13: 1647-1659. PMID: 29490753
- Synthesis, Structure and In Vitro Cytotoxic Activity of Novel Cinchona-Chalcone Hybrids with 1,4-Disubstituted- and 1,5-Disubstituted 1,2,3-Triazole Linkers. | Jernei, T., et al. 2019. Molecules. 24: PMID: 31718009
- Discovery of Novel Sphingosine-1-Phosphate-1 Receptor Agonists for the Treatment of Multiple Sclerosis. | Park, SJ., et al. 2022. J Med Chem. 65: 3539-3562. PMID: 35077170
- Guanidine Derivatives Containing the Chalcone Skeleton Are Potent Antiproliferative Compounds against Human Leukemia Cells. | Estévez-Sarmiento, F., et al. 2022. Int J Mol Sci. 23: PMID: 36555165
- Photochemical cross-linking of elongation factor G to 70-S ribosomes from Escherichia coli by 4-(6-formyl-3-azidophenoxy)butyrimidate. | Maassen, JA. and Möller, W. 1981. Eur J Biochem. 115: 279-85. PMID: 7016534
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Azidobenzaldehyde, 250 mg | sc-503201 | 250 mg | $357.00 | |||
4-Azidobenzaldehyde, 2.5 g | sc-503201A | 2.5 g | $2497.00 |
