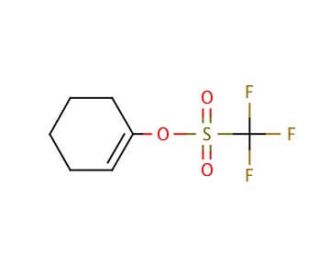
1-Cyclohexenyl trifluoromethanesulfonate (CAS 28075-50-5)
LINKS RÁPIDOS
O trifluorometanossulfonato de 1-ciclohexenilo (1-CHTMS) é um reagente altamente versátil amplamente utilizado na química orgânica sintética, especificamente para a síntese de diversos compostos. A sua importância reside no facto de servir como um bloco de construção fundamental para a síntese de moléculas complexas. A reatividade exibida pelo trifluorometanosulfonato de 1-ciclohexenilo torna-o uma ferramenta inestimável para a síntese de uma vasta gama de compostos. Na ciência dos materiais, o trifluorometanosulfonato de 1-ciclohexenilo actua como catalisador na síntese de polímeros e sistemas de copolímeros. Além disso, no domínio da bioquímica, serve como reagente na síntese de proteínas e péptidos. A elevada reatividade do trifluorometanosulfonato de 1-ciclohexenilo resulta da sua capacidade de formar ligações covalentes com uma gama diversificada de substratos. Isto ocorre quando a molécula de trifluorometanosulfonato de 1-ciclohexenilo doa um par de electrões ao substrato, resultando na formação de uma nova ligação covalente. Subsequentemente, esta ligação covalente sofre clivagem aquando da oxidação do substrato, levando à libertação da molécula de 1-ciclohexenil trifluorometanosulfonato.
1-Cyclohexenyl trifluoromethanesulfonate (CAS 28075-50-5) Referencias
- A trifluorometilação de sulfonatos de vinilo catalisada por paládio. | Cho, EJ. and Buchwald, SL. 2011. Org Lett. 13: 6552-5. PMID: 22111687
- Influência da estrutura do catalisador e das condições de reação nas vias de anti-aminopaladação versus sin-aminopaladação em reacções de carboaminação de alquenos catalisadas por Pd de N-alilsulfamidas. | Fornwald, RM., et al. 2014. Chemistry. 20: 8782-90. PMID: 24938206
- Hidroalkenilação enantioselectiva de olefinas com enol sulfonatos possibilitada pela catálise dupla de hidreto de cobre e paládio. | Schuppe, AW., et al. 2021. J Am Chem Soc. 143: 5330-5335. PMID: 33784090
- Uma síntese estereosselectiva catalisada por CuH e Pd dupla de 1,3-dienes altamente substituídos. | Hou, CJ., et al. 2021. Org Lett. 23: 8816-8821. PMID: 34726414
Informacoes sobre ordens
| Nome do Produto | Numero de Catalogo | UNID | Preco | Qde | FAVORITOS | |
1-Cyclohexenyl trifluoromethanesulfonate, 5 g | sc-224633 | 5 g | $148.00 |
